Chapter 10: Q10-25E-c (page 308)
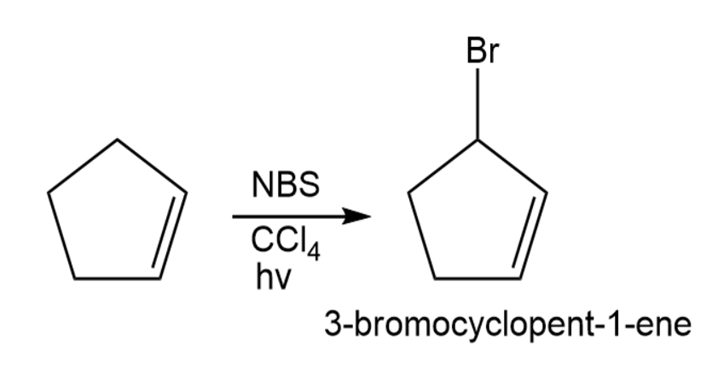
How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (c) 3-Bromocyclopentene.
Short Answer
3-Bromocyclopentene can be formed in the following way:
Learning Materials
Features
Discover
Chapter 10: Q10-25E-c (page 308)
How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (c) 3-Bromocyclopentene.
3-Bromocyclopentene can be formed in the following way:

All the tools & learning materials you need for study success - in one app.
Get started for free
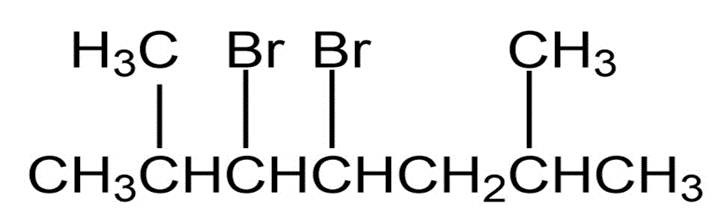
Question: Name the following alkyl halides:
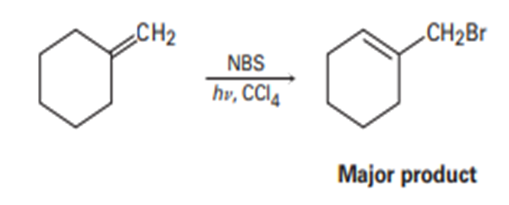
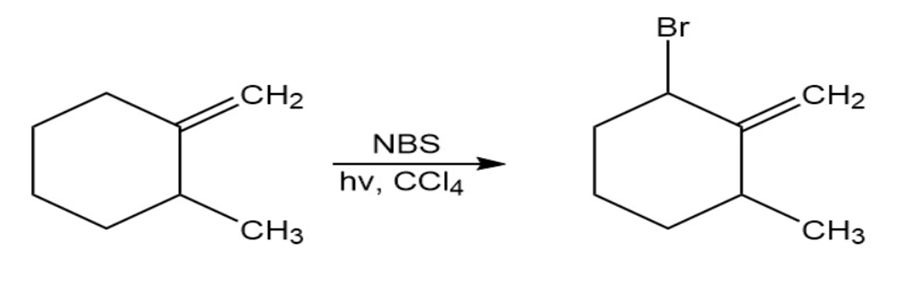
The major product of the reaction of methylenecyclohexane with N-bromosuccinimide is 1-(bromomethyl)cyclohexene. Explain.
Question: The syntheses shown here are unlikely to occur as written. What is
wrong with each?
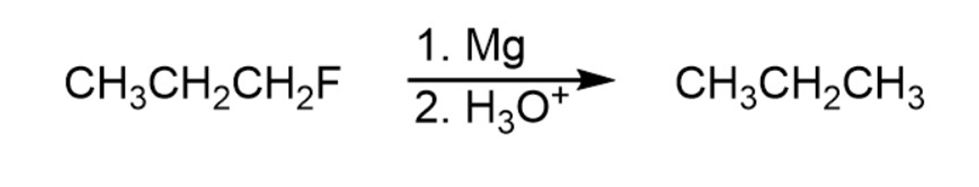
Reaction 1
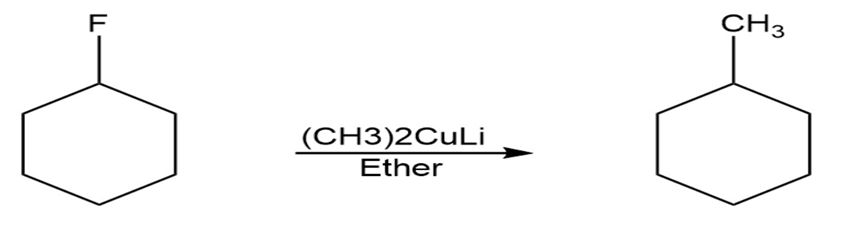
Reaction 2
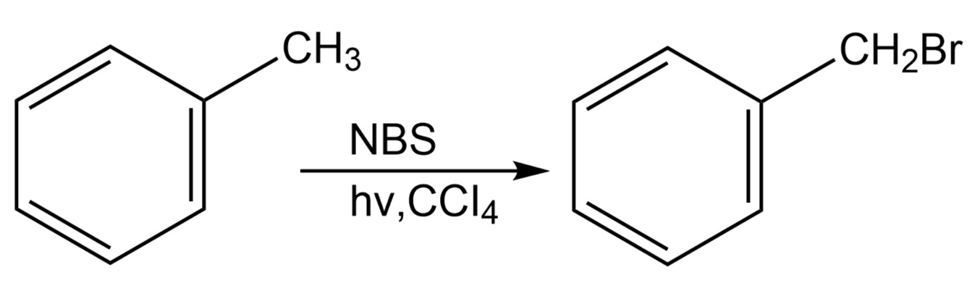
Alkylbenzenes such as toluene (methylbenzene) react with NBS to give
products in which bromine substitution has occurred next to the aromatic ring (the benzylic position). Explain, based on the bond dissociation energies in Table 6-3 on page 170.
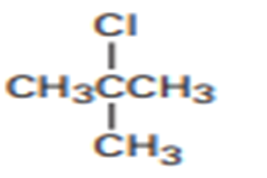
How would you prepare the following alkyl halides from the corresponding alcohols?
What do you think about this solution?
We value your feedback to improve our textbook solutions.