Chapter 10: Q22E-a (page 308)
Question: Name the following alkyl halides:

Short Answer
(a)The name of the compound (a) is 3,4-dibromo-2,6-dimethylheptane.
Learning Materials
Features
Discover
Chapter 10: Q22E-a (page 308)
Question: Name the following alkyl halides:

(a)The name of the compound (a) is 3,4-dibromo-2,6-dimethylheptane.
All the tools & learning materials you need for study success - in one app.
Get started for free
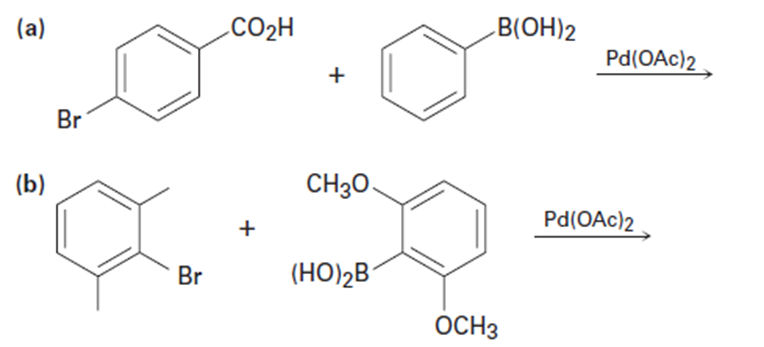
Predict the product and provide the entire catalytic cycle for theSuzuki–Miyaura reactions below.
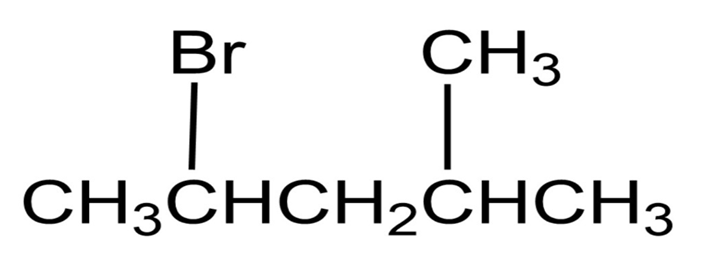
How would you prepare the following alkyl halides from the corresponding alcohols?
Carboxylic acids (RCOOH; =5) are approximately 1011 times more
acidic than alcohols (ROH;=16). In other words, a carboxylate ion
is more stable than an alkoxide ion . Explain, using
resonance.
How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (e) Cyclopentylcyclopentane.
A chemist requires a large amount of 1-bromo-2-pentene as starting material for synthesis and decides to carry out an NBS allylic bromination reaction. What is wrong with the following synthesis plan? What side products would form in addition to the desired product?
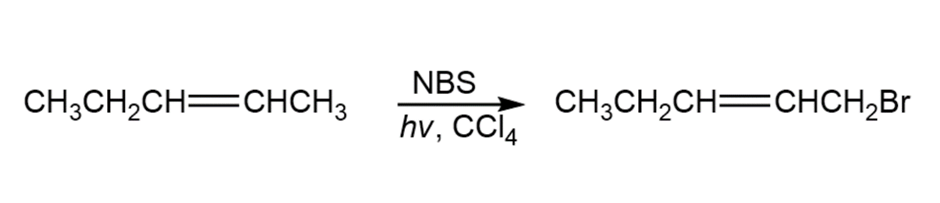
What do you think about this solution?
We value your feedback to improve our textbook solutions.