Chapter 10: Q50 E. (page 308)
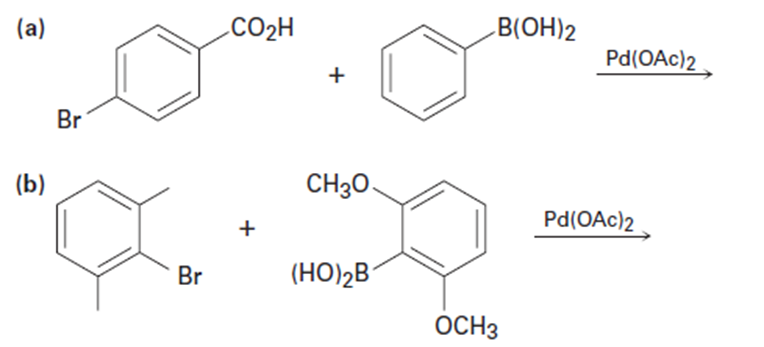
Predict the product and provide the entire catalytic cycle for theSuzuki–Miyaura reactions below.
Short Answer
(a)
(b)
Learning Materials
Features
Discover
Chapter 10: Q50 E. (page 308)
Predict the product and provide the entire catalytic cycle for theSuzuki–Miyaura reactions below.

(a)
(b)
All the tools & learning materials you need for study success - in one app.
Get started for free
Carboxylic acids (RCOOH; =5) are approximately 1011 times more
acidic than alcohols (ROH;=16). In other words, a carboxylate ion
is more stable than an alkoxide ion . Explain, using
resonance.
Name the following alkyl halides:
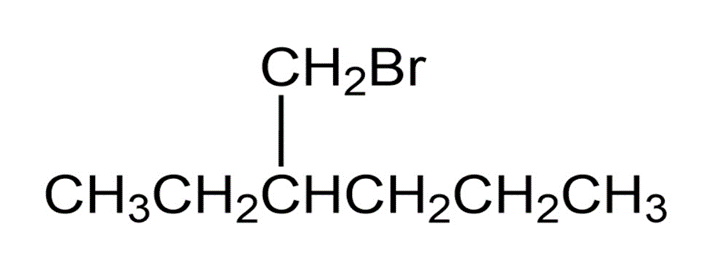
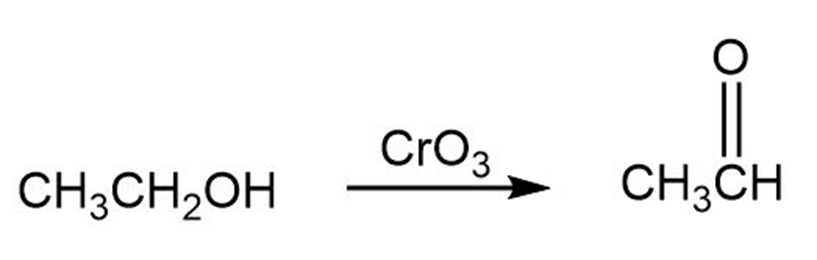
Tell whether each of the following reactions is an oxidation, a reduction, or neither:
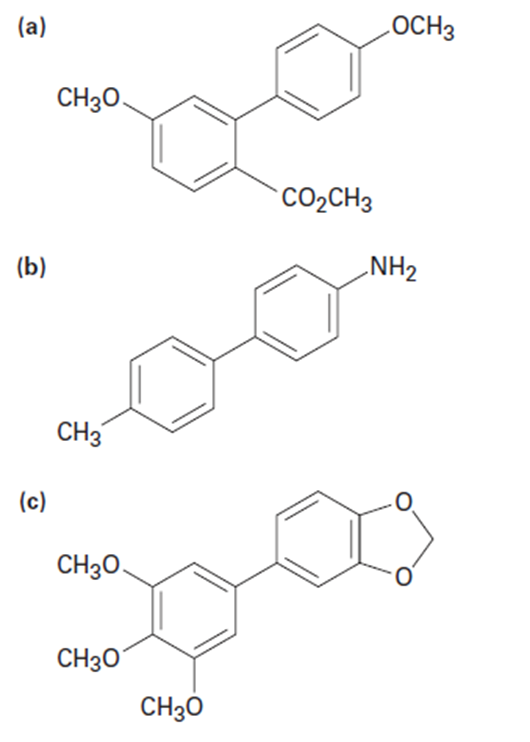
How might you use a Suzuki–Miyaura reaction to prepare the biarylcompounds below? In each case, show the two potential reactionpartners.
Draw structures corresponding to the following IUPAC names: (e) 4-sec-Butyl-2-chlorononane.
What do you think about this solution?
We value your feedback to improve our textbook solutions.