Chapter 10: Q47E (page 308)
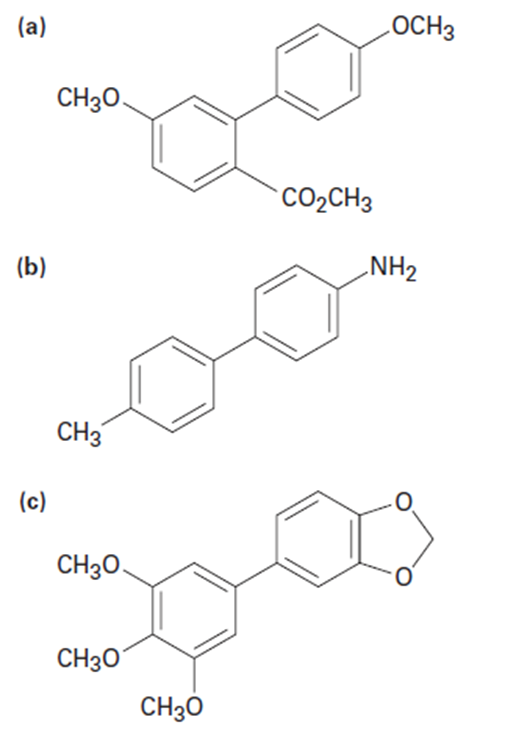
How might you use a Suzuki–Miyaura reaction to prepare the biarylcompounds below? In each case, show the two potential reactionpartners.
Short Answer
(a)

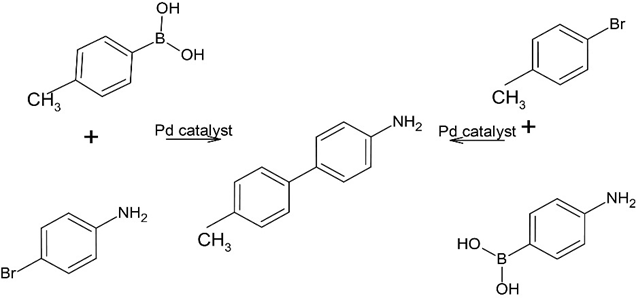
(b)
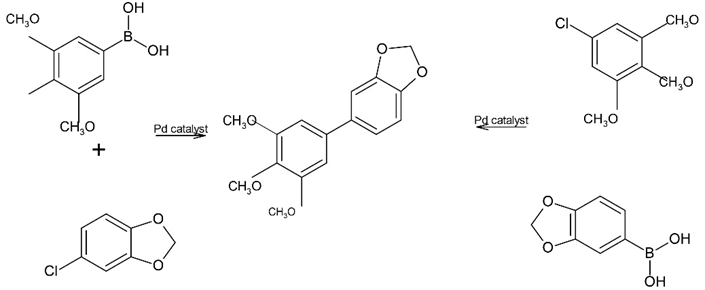
(c)
Learning Materials
Features
Discover
Chapter 10: Q47E (page 308)
How might you use a Suzuki–Miyaura reaction to prepare the biarylcompounds below? In each case, show the two potential reactionpartners.

(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Assume that you have carried out a radical chlorination reaction on
and have isolated (in low yield) . How many stereoisomers of the product are formed, and in what ratio? Are any of the isomers optically active? (See Problem 10-38.)
Question: How might you replace a halogen substituent by a deuterium atom if you wanted to prepare a deuterated compound?

How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (d) Cyclopentanol.
Rank the compounds in each of the following series in order of increasing oxidation level:
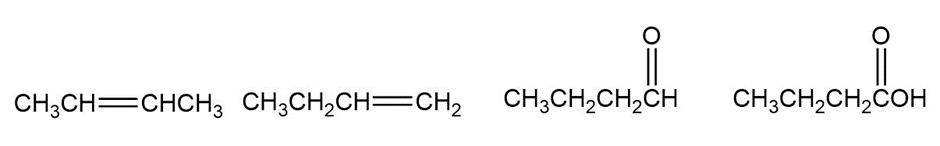
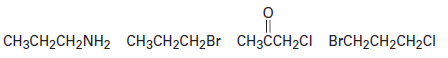
Draw structures corresponding to the following IUPAC names: (a) 2,3-Dichloro-4-methylhexane.
What do you think about this solution?
We value your feedback to improve our textbook solutions.