Chapter 10: Q1P-d (page 290)
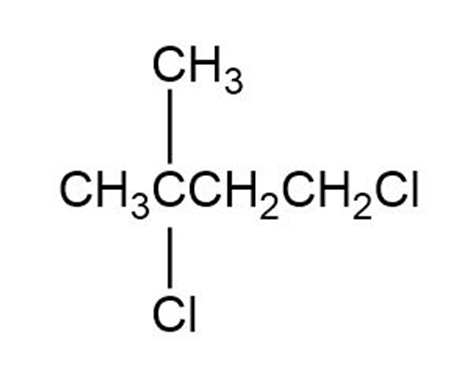
Give IUPAC names for the following alkyl halide (d):
Short Answer
(d)The name of the compound (d) is 1,3-Dichloro-3-Methylbutane.
Learning Materials
Features
Discover
Chapter 10: Q1P-d (page 290)
Give IUPAC names for the following alkyl halide (d):

(d)The name of the compound (d) is 1,3-Dichloro-3-Methylbutane.
All the tools & learning materials you need for study success - in one app.
Get started for free
How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (b) Methylcyclopentane.
Draw resonance structures for the benzyl radical, the intermediate produced in the NBS bromination reaction of toluene (Problem 10-35).
What product(s) would you expect from the reaction of 1,4-hexadiene with NBS? What is the structure of the most stable radical intermediate?
Draw three resonance forms for the cyclohexadienyl radical.
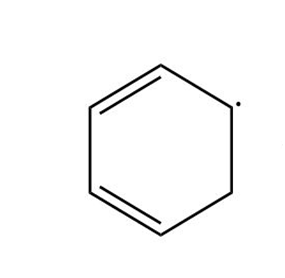
Cyclohexadienyl radical
Question: Assume that you have carried out a radical chlorination reaction on
and have isolated (in low yield) . How many stereoisomers of the product are formed, and in what ratio? Are any of the isomers optically active? (See Problem 10-38.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.