Chapter 10: Q1P-c (page 290)
Give IUPAC names for the following alkyl halide (c):

Short Answer
(c)The name of the compound (c) is 1,5-Dibromo-2,2-Dimethylpentane.
Learning Materials
Features
Discover
Chapter 10: Q1P-c (page 290)
Give IUPAC names for the following alkyl halide (c):

(c)The name of the compound (c) is 1,5-Dibromo-2,2-Dimethylpentane.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Addition of HBr to a double bond with an ethersubstituentoccurs regiospecifically to give a product in which theandare bonded to the same carbon. Draw the two possible carbocation intermediates in this electrophilic addition reaction, and explain usingresonance why the observed product is formed.
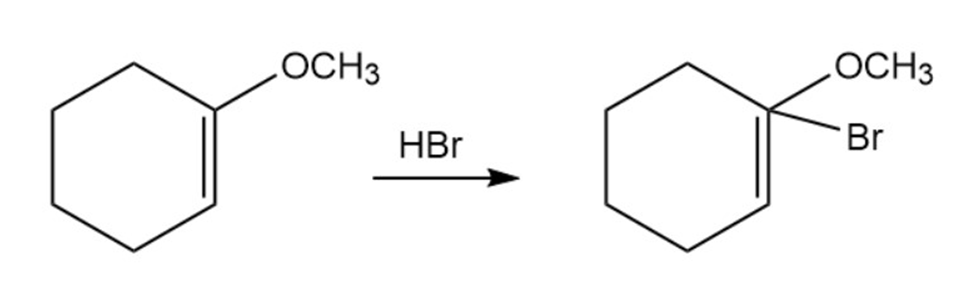
A chemist requires a large amount of 1-bromo-2-pentene as starting material for synthesis and decides to carry out an NBS allylic bromination reaction. What is wrong with the following synthesis plan? What side products would form in addition to the desired product?
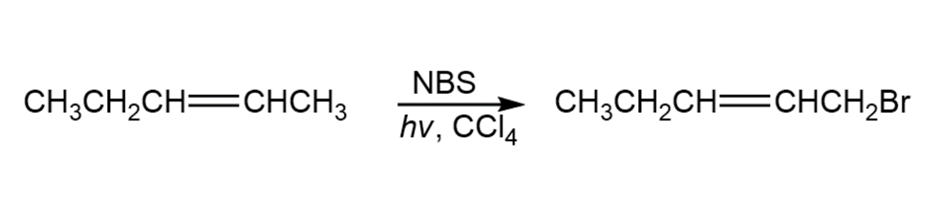
Taking the relative reactivities of 1°, 2°, and 3° hydrogen atoms into account, what product(s) would you expect to obtain from monochlorination of 2-methylbutane? What would the approximate percentage of each product be? (Don’t forget to take into account the number of each kind of hydrogen.)
Alkylbenzenes such as toluene (methylbenzene) react with NBS to give
products in which bromine substitution has occurred next to the aromatic ring (the benzylic position). Explain, based on the bond dissociation energies in Table 6-3 on page 170.

In light of the fact that tertiary alkyl halides undergo spontaneous dissociation to yield a carbocation plus halide ion (see Problem 10-45), propose a mechanism for the following reaction.
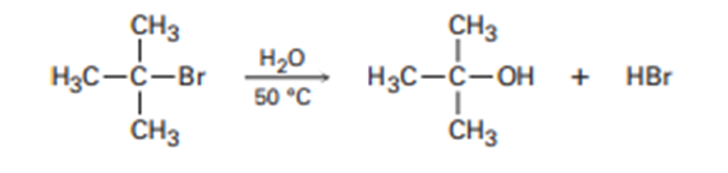
What do you think about this solution?
We value your feedback to improve our textbook solutions.