Chapter 10: Q1P-b (page 290)
Give IUPAC names for the following alkyl halide (b):
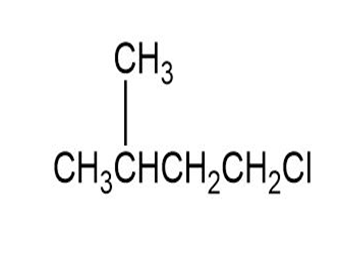
(b)
Short Answer
(b)The name of the compound (b) is 1-Chloro-3-Methylbutane.
Learning Materials
Features
Discover
Chapter 10: Q1P-b (page 290)
Give IUPAC names for the following alkyl halide (b):
(b)

(b)The name of the compound (b) is 1-Chloro-3-Methylbutane.
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw structures corresponding to the following IUPAC names: (c) 3-Iodo-2,2,4,4-tetramethylpentane
A chemist requires a large amount of 1-bromo-2-pentene as starting material for synthesis and decides to carry out an NBS allylic bromination reaction. What is wrong with the following synthesis plan? What side products would form in addition to the desired product?
The relative rate of radical bromination is 1; 82; 1640 for 1°;2°;3°
hydrogens, respectively. Draw all of the monobrominated products that
you might obtain from the radical bromination of the compounds
below. Calculate the relative percentage of each.
(a)Methylcyclobutane
(b)3,3-dimethylpentane
(c)3-methylpentane
What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures of all.

Which of the following compounds have the same oxidation level, and which have different levels?

What do you think about this solution?
We value your feedback to improve our textbook solutions.