Chapter 10: Q32 E (page 308)
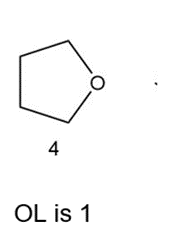
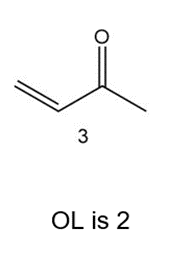
Which of the following compounds have the same oxidation level, and which have different levels?
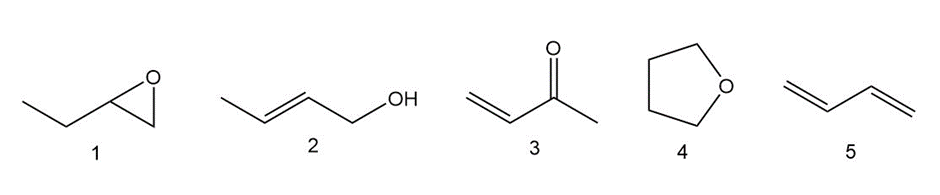
Short Answer
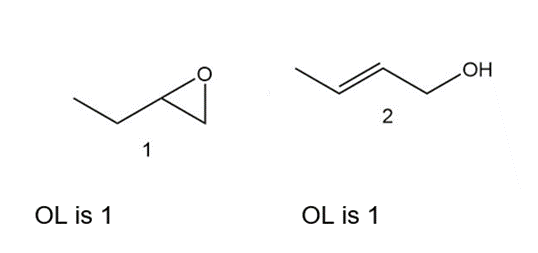
Same oxidation level
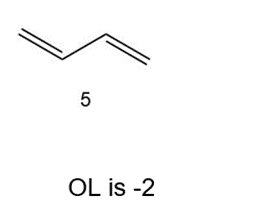
Different oxidation level
Learning Materials
Features
Discover
Chapter 10: Q32 E (page 308)
Which of the following compounds have the same oxidation level, and which have different levels?



Same oxidation level


Different oxidation level
All the tools & learning materials you need for study success - in one app.
Get started for free
Alkyl halides can be reduced to alkanes by a radical reaction with tributyltinhydride, in the presence of light (hv). Propose aradical chain mechanism by which the reaction might occur. The initiationstep is the light-induced homolytic cleavage of the Sn-H bondto yield a tributyltin radical.
Draw structures corresponding to the following IUPAC names: (b) 4-Bromo-4-ethyl-2-methylhexane.
Draw structures corresponding to the following IUPAC names: (f) 1,1-Dibromo-4-tert-butylcyclohexane.
How would you prepare the following alkyl halides from the corresponding alcohols?
How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (c) 3-Bromocyclopentene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.