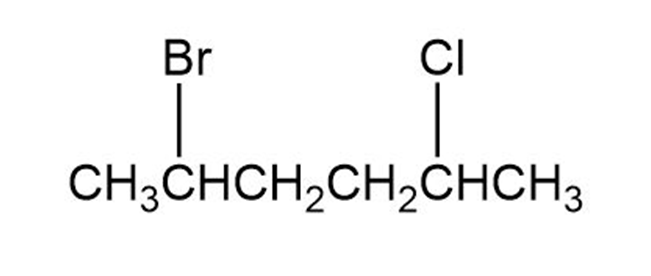
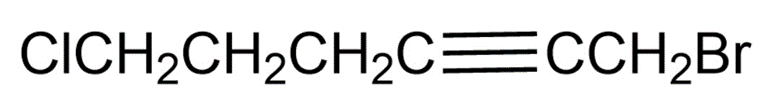Chapter 10: 10-10-21 E (page 308)
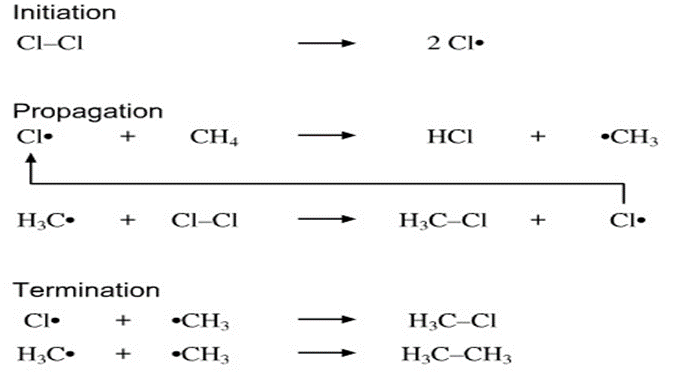
Alkyl halides can be reduced to alkanes by a radical reaction with tributyltinhydride, in the presence of light (hv). Propose aradical chain mechanism by which the reaction might occur. The initiationstep is the light-induced homolytic cleavage of the Sn-H bondto yield a tributyltin radical.
Short Answer
Radical chain mechanism of the reaction is: