Chapter 10: Q 30P (page 308)
What product would you expect from the reaction of 1-phenyl-2-butene with NBS? Explain.
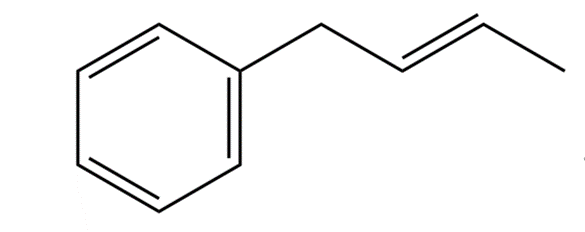
1-phenyl 2-butene
Short Answer
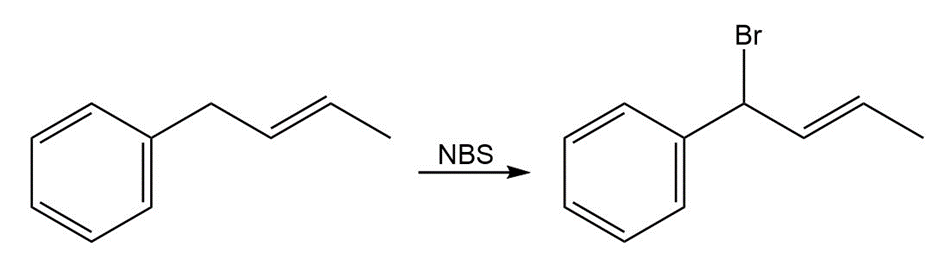
Formation of the desired product
Learning Materials
Features
Discover
Chapter 10: Q 30P (page 308)
What product would you expect from the reaction of 1-phenyl-2-butene with NBS? Explain.

1-phenyl 2-butene

Formation of the desired product
All the tools & learning materials you need for study success - in one app.
Get started for free
How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (b) Methylcyclopentane.
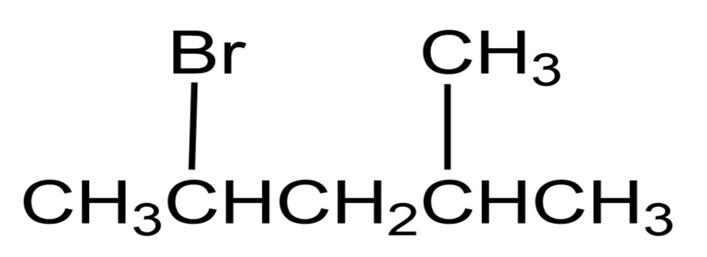
Question: Name the following alkyl halides:
Draw the electron-pushing mechanism for the propagation steps of the allylic bromination reactions below. You may omit NBS in your mechanism, and use Br∙ and Br2.
(b)
Taking the relative reactivities of 1°, 2°, and 3° hydrogen atoms into account, what product(s) would you expect to obtain from monochlorination of 2-methylbutane? What would the approximate percentage of each product be? (Don’t forget to take into account the number of each kind of hydrogen.)
How would you prepare the following alkyl halides from the corresponding alcohols?
What do you think about this solution?
We value your feedback to improve our textbook solutions.