Chapter 10: Q45E (page 308)
Tertiary alkyl halides,, undergo spontaneous dissociation to yield a carbocation,, plus halide ion. Which do you think reacts faster,? Explain.
Short Answer
will react faster.
Learning Materials
Features
Discover
Chapter 10: Q45E (page 308)
Tertiary alkyl halides,, undergo spontaneous dissociation to yield a carbocation,, plus halide ion. Which do you think reacts faster,? Explain.
will react faster.
All the tools & learning materials you need for study success - in one app.
Get started for free
Taking the relative reactivities of 1°, 2°, and 3° hydrogen atoms into account, what product(s) would you expect to obtain from monochlorination of 2-methylbutane? What would the approximate percentage of each product be? (Don’t forget to take into account the number of each kind of hydrogen.)
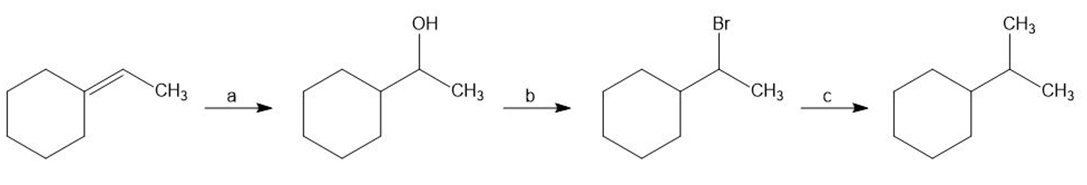
Identify the reagents a–c in the following scheme:
How would you prepare the following compounds, starting with cyclopentene and any other reagents needed? (f) 1,3-Cyclopentadiene.
Draw resonance structures for the following species:
Sort the radicals below from most stable to least stable.
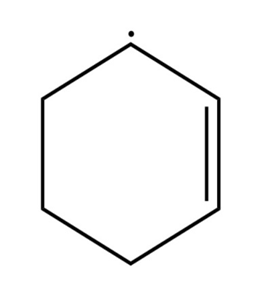
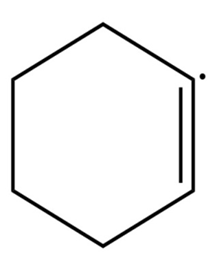
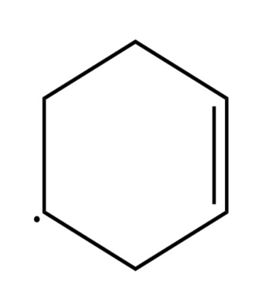
What do you think about this solution?
We value your feedback to improve our textbook solutions.