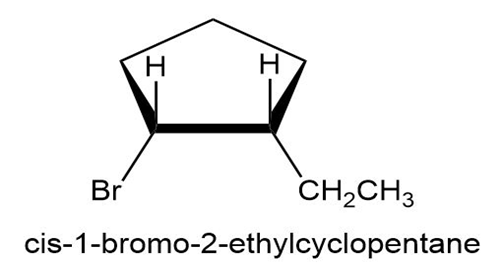
Chapter 10: Q10-23E-d (page 308)
Draw structures corresponding to the following IUPAC names: (d) cis-1-Bromo-2-ethylcyclopentane
Short Answer
(d)The structure of the compound (d) is given below:
Learning Materials
Features
Discover
Chapter 10: Q10-23E-d (page 308)
Draw structures corresponding to the following IUPAC names: (d) cis-1-Bromo-2-ethylcyclopentane
(d)The structure of the compound (d) is given below:

All the tools & learning materials you need for study success - in one app.
Get started for free
Which of the following compounds have the same oxidation level, and which have different levels?
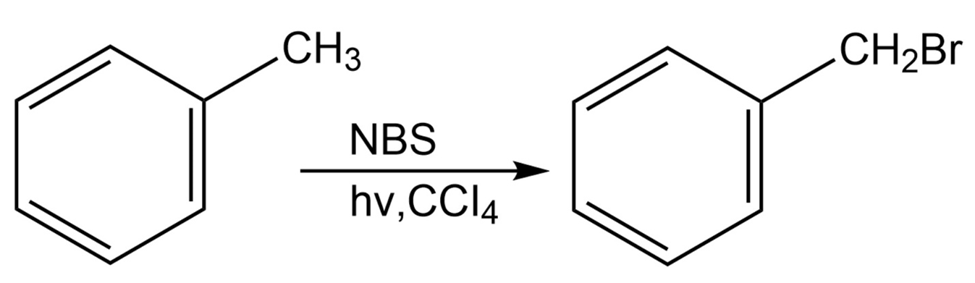
Alkylbenzenes such as toluene (methylbenzene) react with NBS to give
products in which bromine substitution has occurred next to the aromatic ring (the benzylic position). Explain, based on the bond dissociation energies in Table 6-3 on page 170.
Draw and name all of the monochlorination products that you might obtain from the radical chlorination of the compounds below. Which of the products is chiral? Are any of the products optically active?
(a) 2-methylbutane
(b) Methylcyclopropane
(c) 2,2-dimethylpentane
Draw the electron-pushing mechanism for the propagation steps of the allylic bromination reactions below. You may omit NBS in your mechanism, and use Br∙ and Br2.
(b)
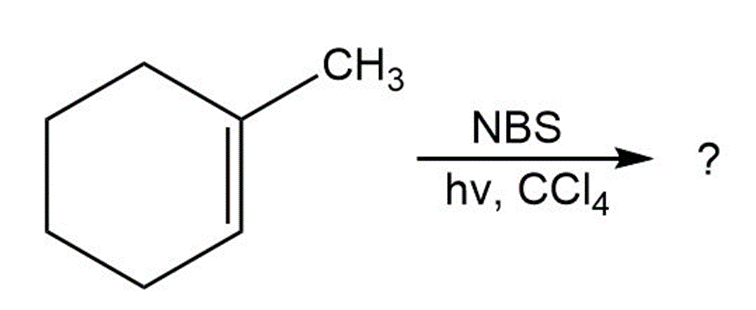
What product(s) would you expect from the reaction of 1-methyl cyclohexene with NBS? Would you use this reaction as part of a synthesis?
What do you think about this solution?
We value your feedback to improve our textbook solutions.