Chapter 12: Q51E (page 354)
Draw the structure of an alkene that yields only acetone, , on ozonolysis followed by treatment with Zn.
Short Answer
Learning Materials
Features
Discover
Chapter 12: Q51E (page 354)
Draw the structure of an alkene that yields only acetone, , on ozonolysis followed by treatment with Zn.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How might you use IR spectroscopy to distinguish between the following pairs of isomers?
Question: What functional groups might the following molecules contain?
(a) A compound with a strong absorption at 1710
(b) A compound with a strong absorption at 1540
(c) A compound with strong absorptions at 1720 and 2500 to 3100
Question: How could you use infrared spectroscopy to distinguish between the
following pairs of isomers?
(a)
(b)
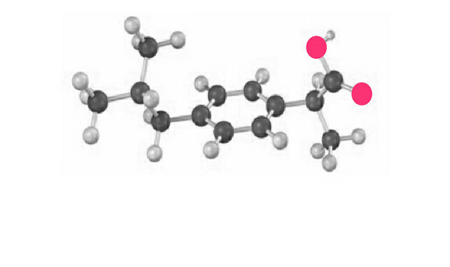
The following model is a representation of ibuprofen, a common over-the-counter pain reliever. Indicate the positions of the multiple bonds, and draw a skeletal structure (gray 5 C, red 5 O, ivory 5 H).
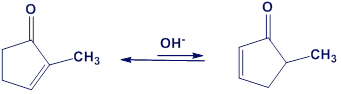
One consequence of the base-catalyzed isomerization of unsaturated ketones described in Problem 22-55 is that 2-substituted 2-cyclopentenones can be interconverted with 5-substituted 2-cyclopentenones. Propose a mechanism for this isomerization.
What do you think about this solution?
We value your feedback to improve our textbook solutions.