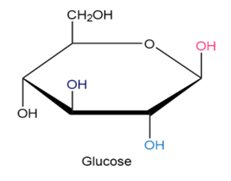
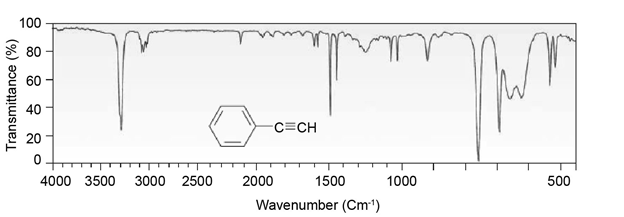
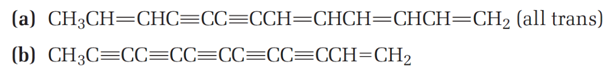Chapter 12: Q27E (page 354)
One consequence of the base-catalyzed isomerization of unsaturated ketones described in Problem 22-55 is that 2-substituted 2-cyclopentenones can be interconverted with 5-substituted 2-cyclopentenones. Propose a mechanism for this isomerization.
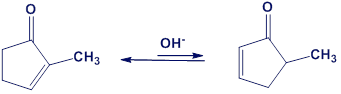
Short Answer
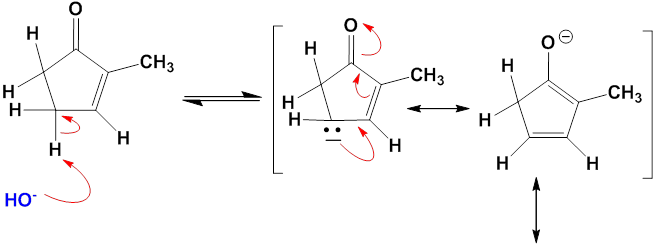
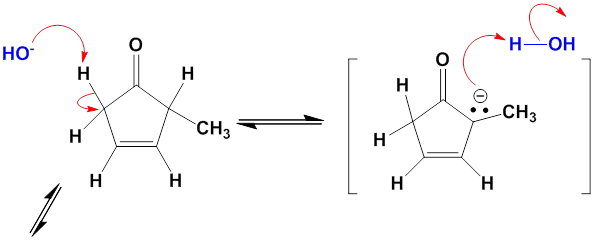
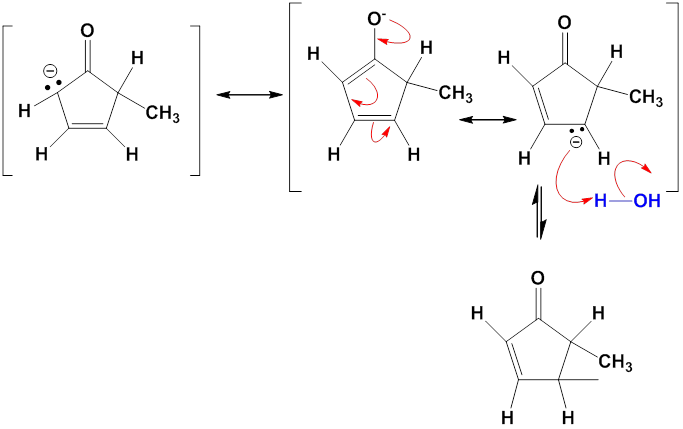
Mechanism
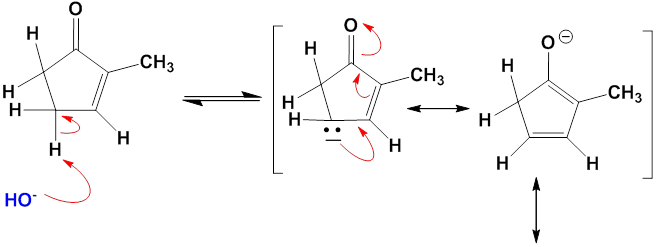
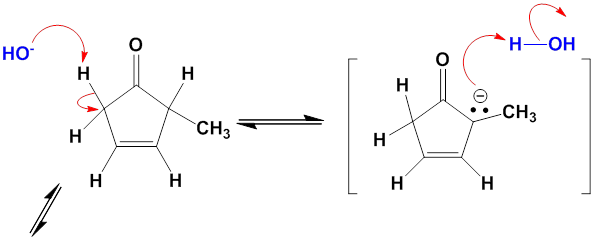
 Mechanism
Mechanism