Chapter 12: Q28E (page 354)
The following two hydrocarbons have been isolated from various plants in the sunflower family. Name them according to IUPAC rules.
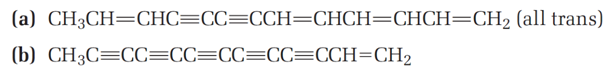
Short Answer
The structure of the given compounds are:
(a)
(b)
Learning Materials
Features
Discover
Chapter 12: Q28E (page 354)
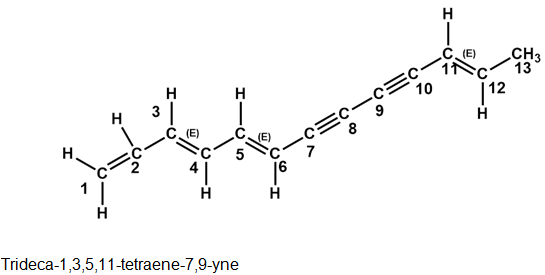
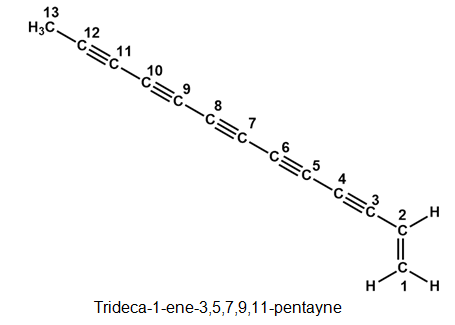
The following two hydrocarbons have been isolated from various plants in the sunflower family. Name them according to IUPAC rules.
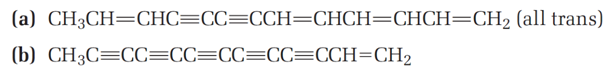
The structure of the given compounds are:
(a)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
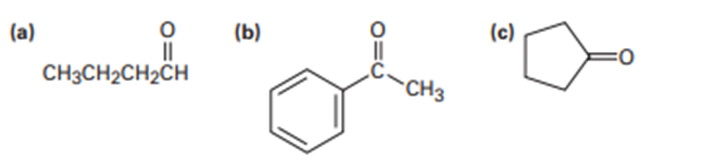
Predict the aldol reaction product of the following compounds:
The amount of energy required to spin-flip a nucleus depends both on the strength of the external magnetic field and on the nucleus. At a field strength
of 4.7 T, rf energy of 200 MHz is required to bring a nucleus into resonance,
but energy of only 187 MHz will bring a nucleus into resonance. Calculate
the amount of energy required to spin-flip a nucleus. Is this amount greater
or less than that required to spin-flip a nucleus?
At what approximate positions might the following compounds show
IR absorptions?
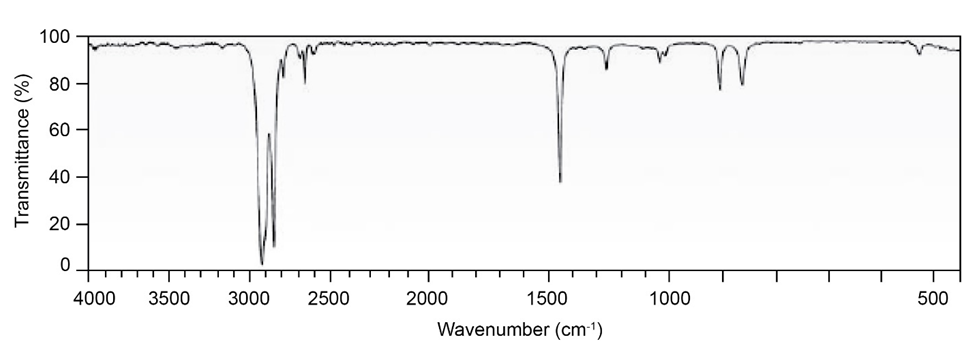
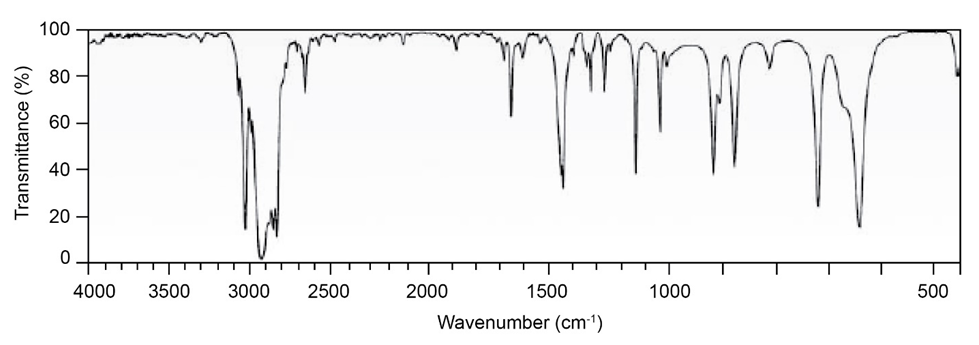
Question: Two infrared spectra are shown. One is the spectrum of cyclohexane,
and the other is the spectrum of cyclohexene. Identify them, and
explain your answer.
(a)
(b)
Question: What functional groups might the following molecules contain?
(a) A compound with a strong absorption at 1710
(b) A compound with a strong absorption at 1540
(c) A compound with strong absorptions at 1720 and 2500 to 3100
What do you think about this solution?
We value your feedback to improve our textbook solutions.