Chapter 12: Q 50 E (page 385)
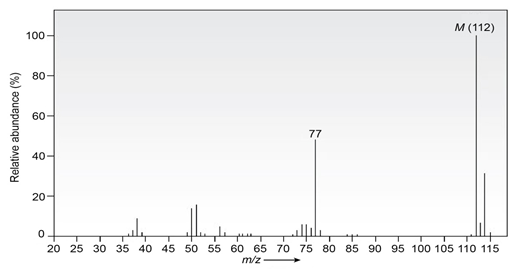
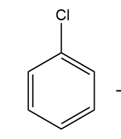
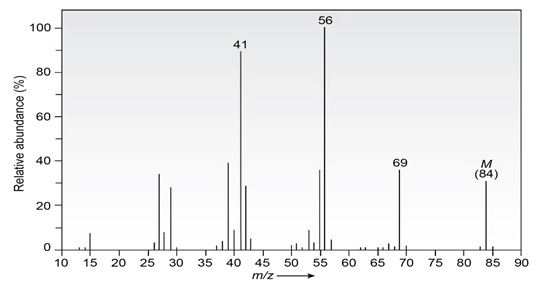
Question: The infrared spectrum of the compound with the mass spectrum shown below has strong absorbances at 1584, 1478, and . Propose astructure consistent with the data.
Short Answer
Learning Materials
Features
Discover
Chapter 12: Q 50 E (page 385)
Question: The infrared spectrum of the compound with the mass spectrum shown below has strong absorbances at 1584, 1478, and . Propose astructure consistent with the data.


All the tools & learning materials you need for study success - in one app.
Get started for free
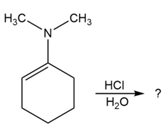
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
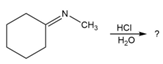
b.
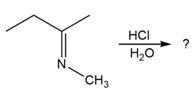
c.
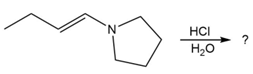
d.
Question: Nitriles,, undergo a hydrolysis reaction when heated withaqueous acid. What is the structure of the compound produced by hydrolysis of propanenitrile, , if it has IR absorptions from and at , and has ?
Question: At what approximate positions might the following compounds showIR absorptions?
(e)
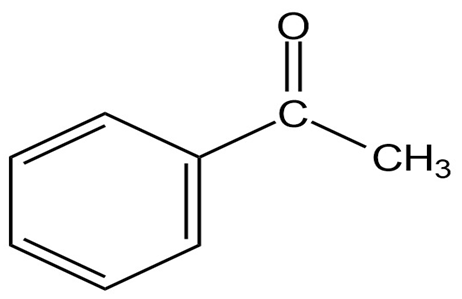
Question: The infrared spectrum of the compound with the mass spectrum shown below has a medium-intensity peak at about . There is also aC-H out-of-plane bending peak near . Propose a structure consistent with the data.
Question: How might you use IR spectroscopy to distinguish between the following pairs of isomers?
What do you think about this solution?
We value your feedback to improve our textbook solutions.