Chapter 12: Q35e E (page 385)
Question: At what approximate positions might the following compounds showIR absorptions?
(e)
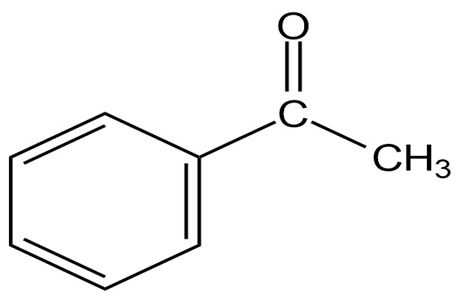
Short Answer
(e) Absorption of ketone next to aromatic ring at 1690, and aromatic ring at 3030.
Learning Materials
Features
Discover
Chapter 12: Q35e E (page 385)
Question: At what approximate positions might the following compounds showIR absorptions?
(e)

(e) Absorption of ketone next to aromatic ring at 1690, and aromatic ring at 3030.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Halogenated compounds are particularly easy to identify by their mass
spectra because both chlorine and bromine occur naturally as mixtures
of two abundant isotopes. Recall that chlorine occurs as 35Cl (75.8%)
and 37C (24.2%); and bromine occurs as 79Br (50.7%) and 81Br (49.3%).
At what masses do the molecular ions occur for the following formulas?
What are the relative percentages of each molecular ion?
(a)Bromomethane, CH3Br
(b)1-Chlorohexane, C6H13Cl
Draw the structure of an alkene that yields only acetone, , on ozonolysis followed by treatment with Zn.
Question: How could you use infrared spectroscopy to distinguish between the
following pairs of isomers?
(a)
(b)
Which of the following bases could be used to deprotonate 1-butyne?
(a) KOH
Question: Propose structures for compounds that fit the following mass-spectral data:
(a) A hydrocarbon with
(b) A hydrocarbon with
(c) A hydrocarbon with
What do you think about this solution?
We value your feedback to improve our textbook solutions.