Chapter 12: Q35 E (page 354)
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
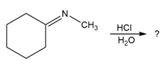
b.
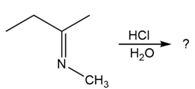
c.
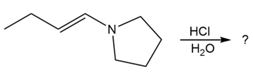
d.
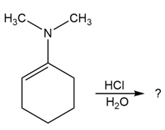
Short Answer
a.
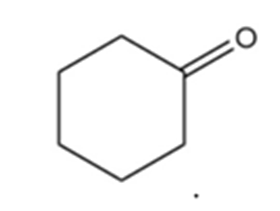
b.
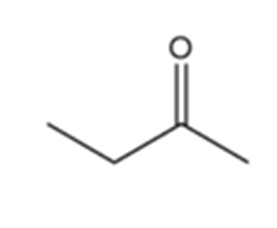
c.
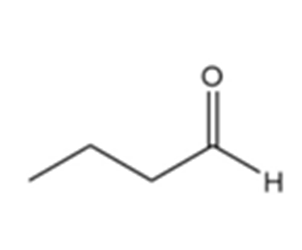
d.
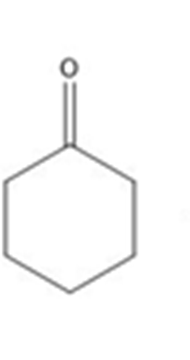
Learning Materials
Features
Discover
Chapter 12: Q35 E (page 354)
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?

b.

c.

d.

a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
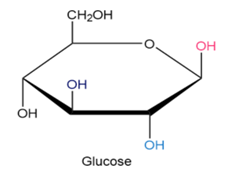
Identify each pair of relationships among the groups in glucose (pink–blue, pink–dark blue, pink–black, blue–dark blue, blue-black, dark blue-black) as cis or trans.
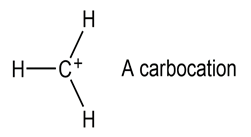
Most stable organic species have tetravalent carbon atoms, but species with trivalent carbon atoms also exist. Carbocations are one such class of compounds.
(a) How many valence electrons does the positively charged carbon atom have?
(b) What hybridization do you expect this carbon atom to have?
(c) What geometry is the carbocation likely to have?
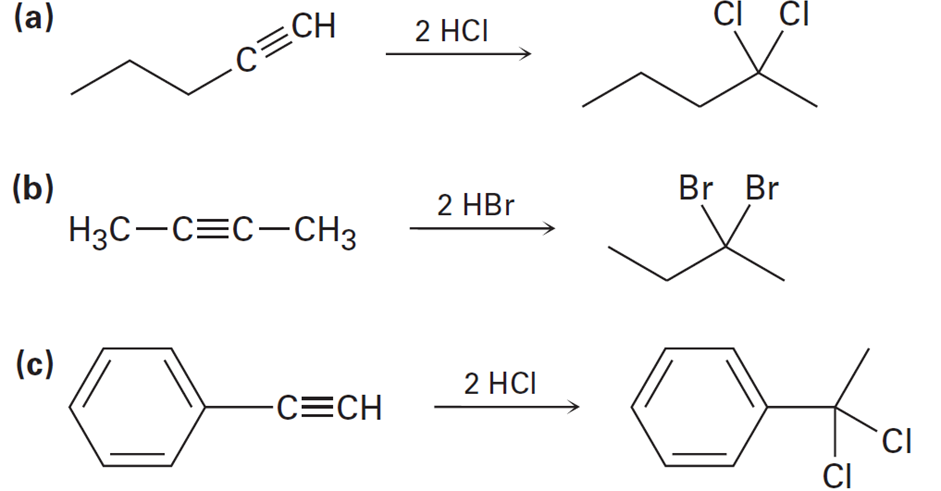
Assuming that strong acids add to alkynes in the same manner as they add to alkenes, propose a mechanism for each of the following reactions:
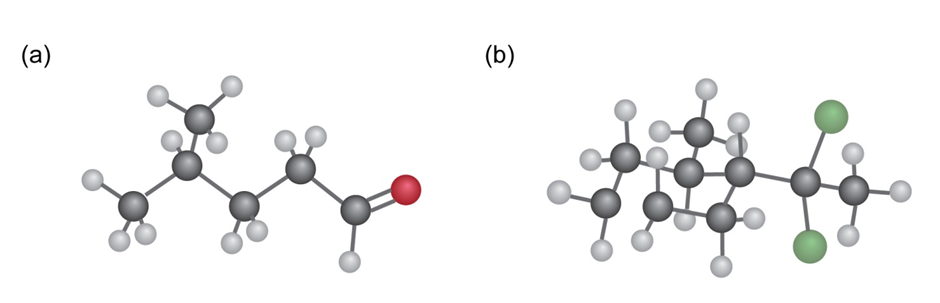
From what alkyne might each of the following substances have been made? (Green = Cl.)
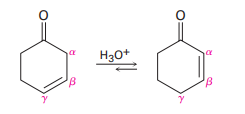
Nonconjugated ,-unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated ,-unsaturated isomers. Propose a mechanism for this isomerization.
What do you think about this solution?
We value your feedback to improve our textbook solutions.