Chapter 12: Q19E (page 354)
Assuming that strong acids add to alkynes in the same manner as they add to alkenes, propose a mechanism for each of the following reactions:
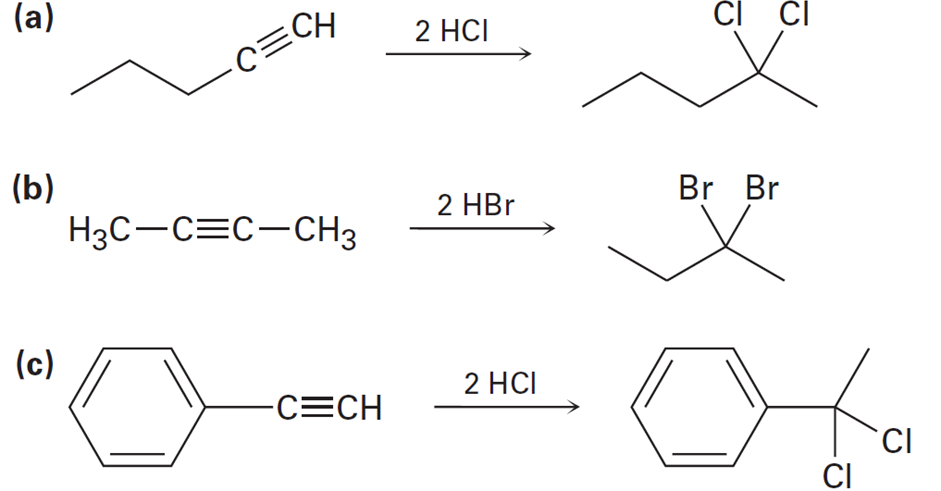
Short Answer
The mechanisms of the reactions are given below.
a).
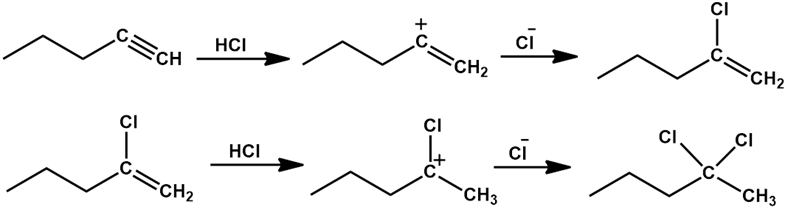
b).
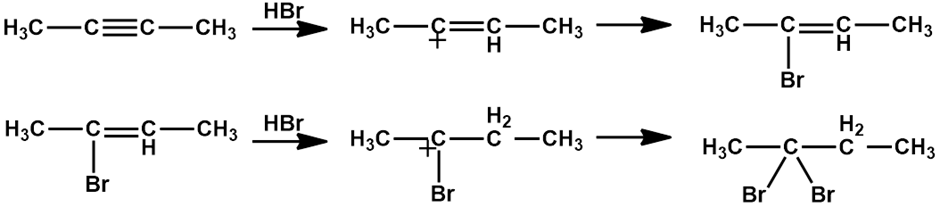
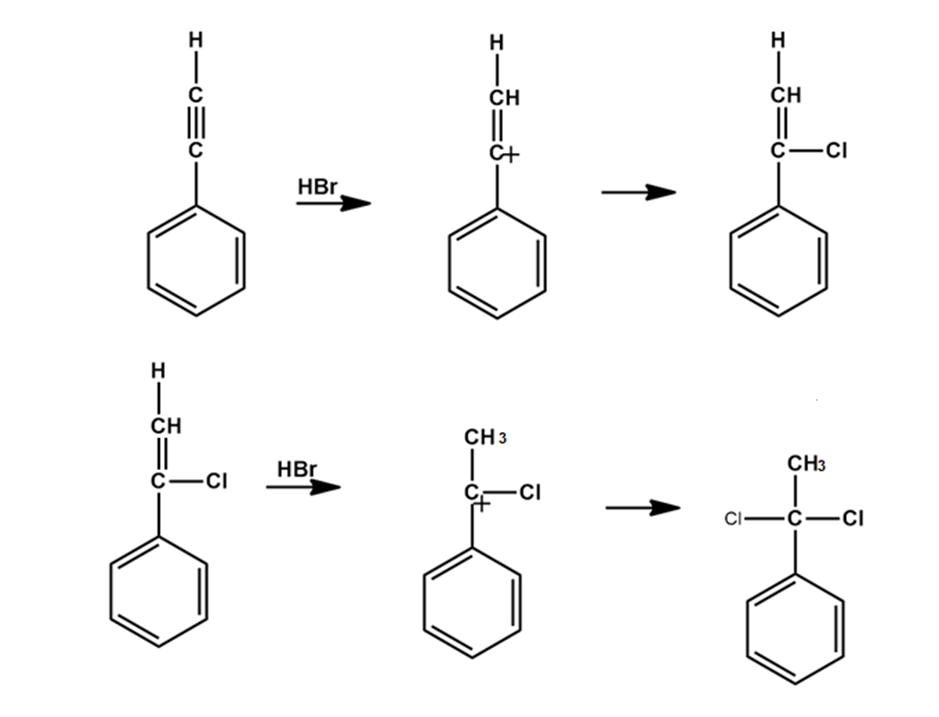
c).
Learning Materials
Features
Discover
Chapter 12: Q19E (page 354)
Assuming that strong acids add to alkynes in the same manner as they add to alkenes, propose a mechanism for each of the following reactions:

The mechanisms of the reactions are given below.
a).

b).

c).

All the tools & learning materials you need for study success - in one app.
Get started for free
Ketones undergo a reduction when treated with sodium borohydride, NaBH4
. What is the structure of the compound produced by reaction
of 2-butanone with NaBH4 if it has an IR absorption at 3400 cm-1 and M+ = 74in the mass spectrum?
At what approximate positions might the following compounds showIR absorptions?
(d)
Most stable organic species have tetravalent carbon atoms, but species with trivalent carbon atoms also exist. Carbocations are one such class of compounds.
(a) How many valence electrons does the positively charged carbon atom have?
(b) What hybridization do you expect this carbon atom to have?
(c) What geometry is the carbocation likely to have?
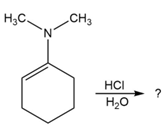
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
b.
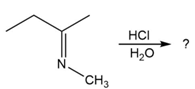
c.
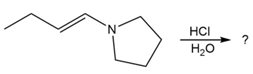
d.
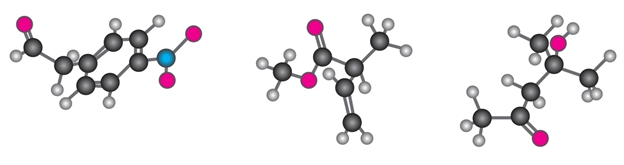
Question: Where might the following compound have IR absorptions?
What do you think about this solution?
We value your feedback to improve our textbook solutions.