Chapter 12: Q 12 P (page 385)
Question: Where might the following compound have IR absorptions?
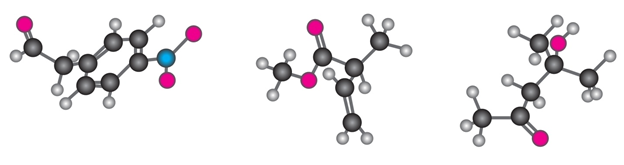
Short Answer
Absorption bands seen at 2210-2260 , 1690 and 1640-1680 .
Learning Materials
Features
Discover
Chapter 12: Q 12 P (page 385)
Question: Where might the following compound have IR absorptions?

Absorption bands seen at 2210-2260 , 1690 and 1640-1680 .
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Halogenated compounds are particularly easy to identify by their mass
spectra because both chlorine and bromine occur naturally as mixtures
of two abundant isotopes. Recall that chlorine occurs as 35Cl (75.8%)
and 37C (24.2%); and bromine occurs as 79Br (50.7%) and 81Br (49.3%).
At what masses do the molecular ions occur for the following formulas?
What are the relative percentages of each molecular ion?
(a)Bromomethane, CH3Br
(b)1-Chlorohexane, C6H13Cl
One consequence of the base-catalyzed isomerization of unsaturated ketones described in Problem 22-55 is that 2-substituted 2-cyclopentenones can be interconverted with 5-substituted 2-cyclopentenones. Propose a mechanism for this isomerization.
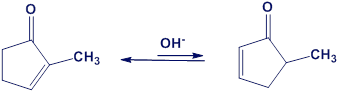
Question: How could you use infrared spectroscopy to distinguish between the
following pairs of isomers?
(a)
(b)
Question: Why do you suppose accidental overlap of signals is much more common in1H NMR than in13C NMR?
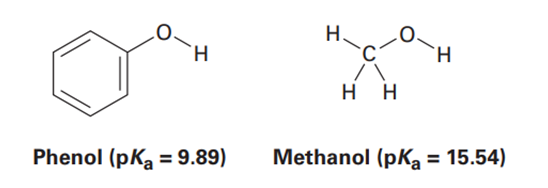
Phenol,, is a stronger acid than methanol, , even though both contain an OH bond. Draw the structures of the anions resulting from loss of from phenol and methanol, and use resonance structures to explain the difference in acidity.
What do you think about this solution?
We value your feedback to improve our textbook solutions.