Chapter 12: Q18E (page 385)
In light of the nitrogen rule mentioned in Problem 12-17, what is themolecular formula of pyridine, M+=79?
Short Answer
The molecular formula of pyridine, M+= 79 is.
Learning Materials
Features
Discover
Chapter 12: Q18E (page 385)
In light of the nitrogen rule mentioned in Problem 12-17, what is themolecular formula of pyridine, M+=79?
The molecular formula of pyridine, M+= 79 is.
All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
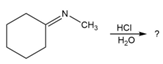
b.
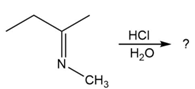
c.
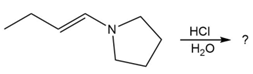
d.
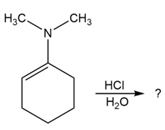
Question: At what approximate positions might the following compounds show
IR absorptions?
(a)
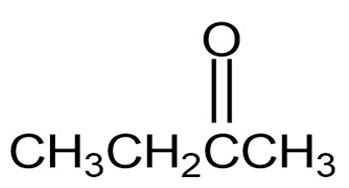
Draw structures corresponding to the following IUPAC names:
a)cis-1,2-Cyclohexanedicarboxylic acid
b) Hepatanedioic acid
c) 2-Hexen-4-ynoic acid
d) 4-Ethyl-2-propyloctanoic acid
e) 3-chlorophthalic acid
f) Triphenylacetic acid
g) 2-Cyclobutenecarbonitrile
h) m-Benzoylbenzonitrile
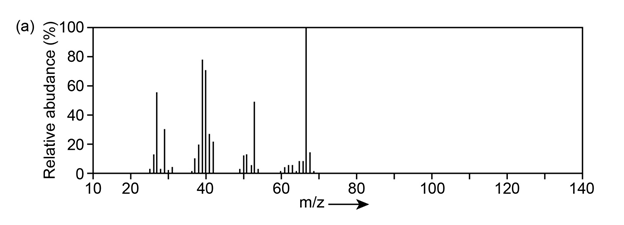
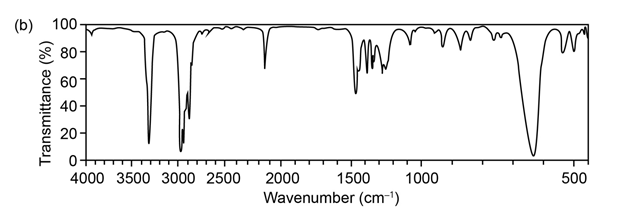
Question:The mass spectrum (a) and the infrared spectrum (b) of an unknown hydrocarbon are shown. Propose as many structures as you can.
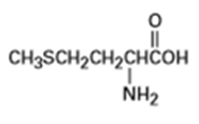
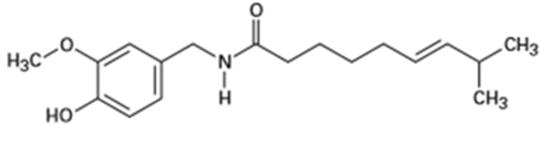
Identify the functional groups in each of the following molecules:
(a) Methionine, an amino acid:
(b)Ibuprofen, a pain reliever:
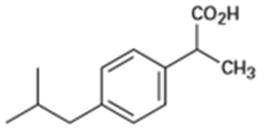
(c) Capsaicin, the pungent substance in chili peppers:
What do you think about this solution?
We value your feedback to improve our textbook solutions.