Chapter 12: 35a E (page 385)
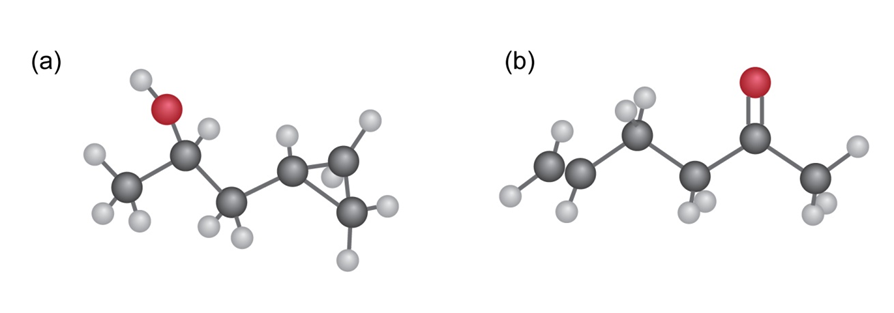
Question: At what approximate positions might the following compounds show
IR absorptions?
(a)
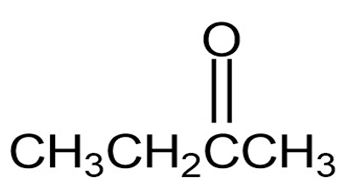
Short Answer
(a) It shows a distinguished absorption due to ketone at 1735.
Learning Materials
Features
Discover
Chapter 12: 35a E (page 385)
Question: At what approximate positions might the following compounds show
IR absorptions?
(a)

(a) It shows a distinguished absorption due to ketone at 1735.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Grignard reagents undergo a general and very useful reaction with
ketones. Methylmagnesium bromide, for example, reacts with cyclohexanoneto yield a product with the formula . What is the structure of this product if it has an IR absorption at ?
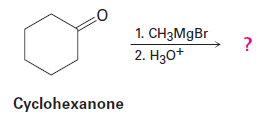
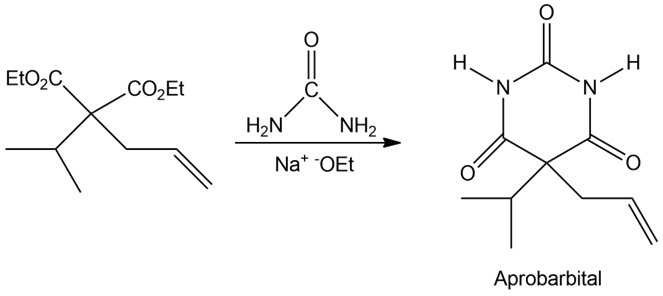
Aprobarbital, a barbiturate once used in treating insomnia, is synthesized in three steps from diethyl malonate. Show how you would synthesize the necessary dialkylated intermediate, and then propose a mechanism for the reaction of this intermediate with urea to give aprobarbital.
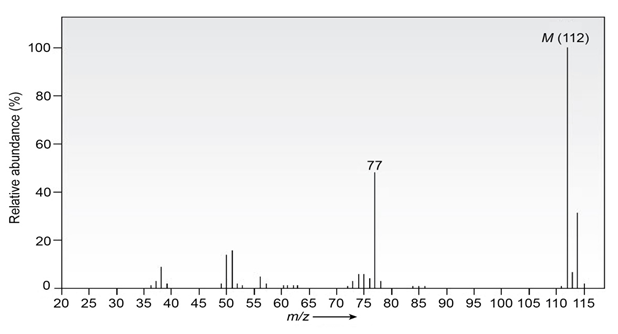
The infrared spectrum of the compound with the mass spectrum shown below has strong absorbances at 1584, 1478, and 1446 cm-1 . Propose a
structure consistent with the data.
How would you prepare the following substances, starting from any
compounds having four carbons or fewer?
Which of the following bases could be used to deprotonate 1-butyne?
(a) KOH
What do you think about this solution?
We value your feedback to improve our textbook solutions.