Chapter 12: 35b E (page 385)
Question: At what approximate positions might the following compounds show
IR absorptions?
(b)
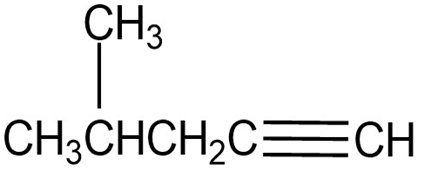
Short Answer
(b) It shows two distinguished absorption due to alkyne at 2100-2600 and at 3300.
Learning Materials
Features
Discover
Chapter 12: 35b E (page 385)
Question: At what approximate positions might the following compounds show
IR absorptions?
(b)
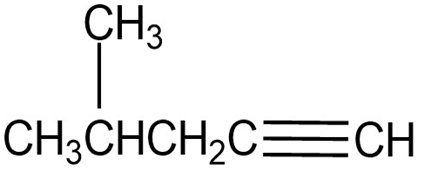
(b) It shows two distinguished absorption due to alkyne at 2100-2600 and at 3300.
All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the products for the reactions in Problem 8-26 if each were run in DMSO with water. Show the complete mechanism including appropriate regiochemistry and stereochemistry.
Nicotine is a diamino compound isolated from dried tobaccoleaves. Nicotinehas two rings and=162.1157 by high-resolution mass spectrometry. Give a molecular formula for nicotine, and calculate the number of double bonds.
Question: Why do you suppose accidental overlap of signals is much more common in1H NMR than in13C NMR?
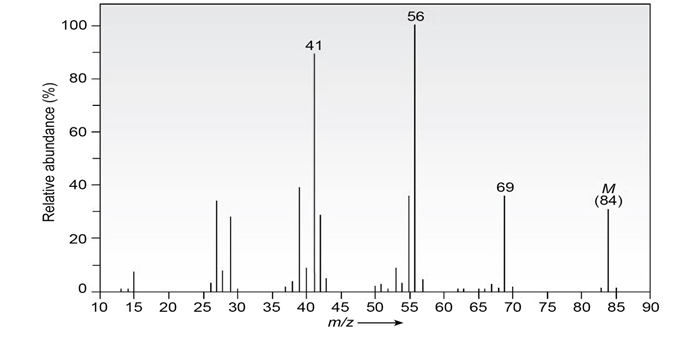
The infrared spectrum of the compound with the mass spectrum shown below has a medium-intensity peak at about 1650cm-1. There is also a
C-H out-of-plane bending peak near 880cm-1 . Propose a structure consistent with the data.
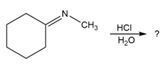
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
b.
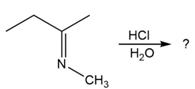
c.
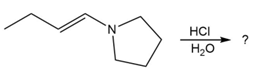
d.
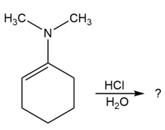
What do you think about this solution?
We value your feedback to improve our textbook solutions.