Chapter 12: Q12-35d E (page 385)
At what approximate positions might the following compounds showIR absorptions?
(d)
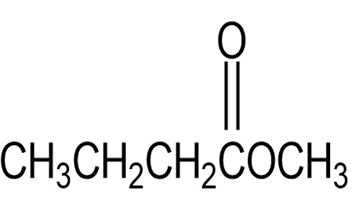
Short Answer
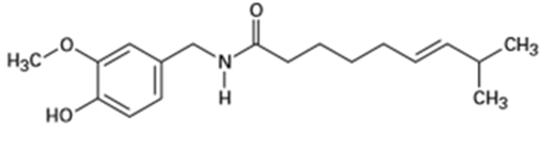
(d) It shows one distinguishing absorption due to ester at 1735.
Learning Materials
Features
Discover
Chapter 12: Q12-35d E (page 385)
At what approximate positions might the following compounds showIR absorptions?
(d)

(d) It shows one distinguishing absorption due to ester at 1735.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: At what approximate positions might the following compounds show
IR absorptions?
(b)
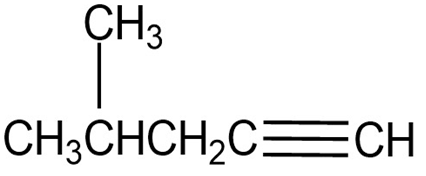
Question: The infrared spectrum of the compound with the mass spectrum shown below lacks any significant absorption above . There is a prominent peak near and another strong peak near .Propose a structure consistent with the data.

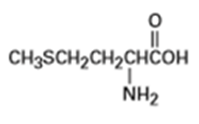
Identify the functional groups in each of the following molecules:
(a) Methionine, an amino acid:
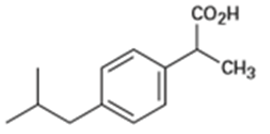
(b)Ibuprofen, a pain reliever:
(c) Capsaicin, the pungent substance in chili peppers:
Question: The nitrogen ruleof mass spectrometry says that a compound containing an odd number of nitrogen’s has an odd-numbered molecular ion.
Conversely, a compound containing an even number of nitrogen’s has
an even-numbered M+peak. Explain.
Question: Two infrared spectra are shown. One is the spectrum of cyclohexane,
and the other is the spectrum of cyclohexene. Identify them, and
explain your answer.
(a)

(b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.