Chapter 12: Q 14 P (page 385)
Question: Propose structures for compounds that fit the following mass-spectral data:
(a) A hydrocarbon with
(b) A hydrocarbon with
(c) A hydrocarbon with
Short Answer
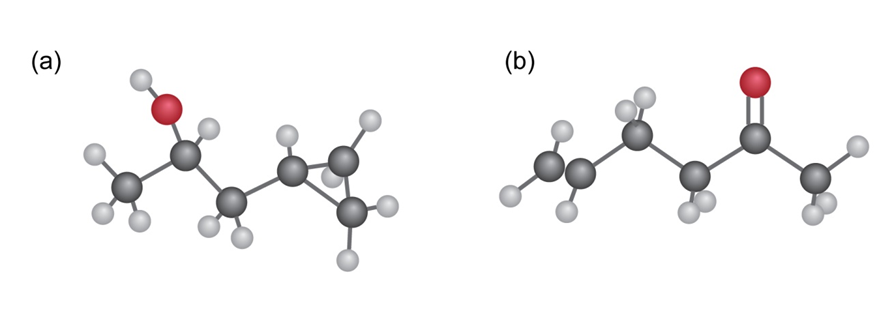
Possible structures of molecule:
a)
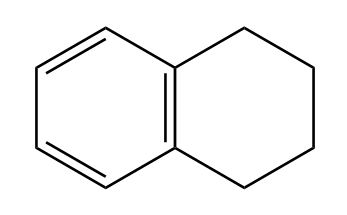
b)
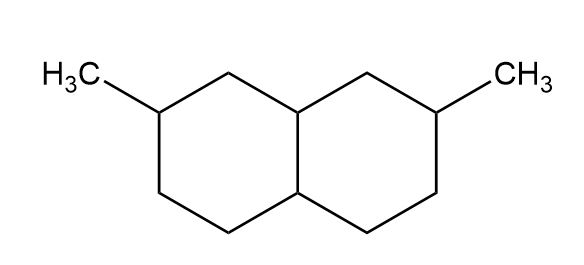
c)
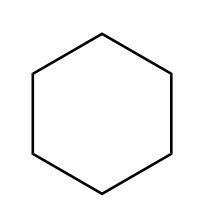
Learning Materials
Features
Discover
Chapter 12: Q 14 P (page 385)
Question: Propose structures for compounds that fit the following mass-spectral data:
(a) A hydrocarbon with
(b) A hydrocarbon with
(c) A hydrocarbon with
Possible structures of molecule:
a)

b)

c)

All the tools & learning materials you need for study success - in one app.
Get started for free
The infrared spectrum of the compound with the mass spectrum shown below has a medium-intensity peak at about 1650cm-1. There is also a
C-H out-of-plane bending peak near 880cm-1 . Propose a structure consistent with the data.
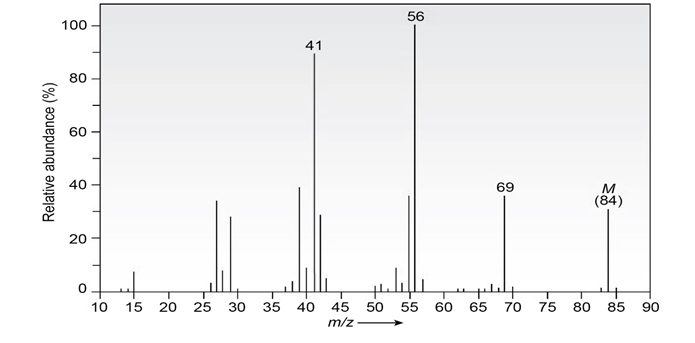
How would you prepare the following substances, starting from any
compounds having four carbons or fewer?
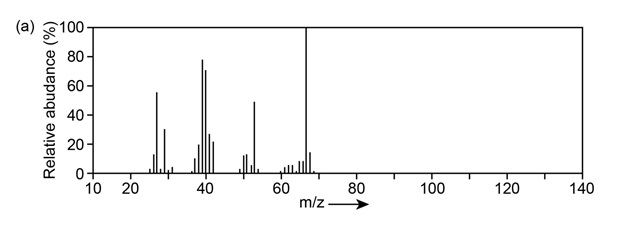
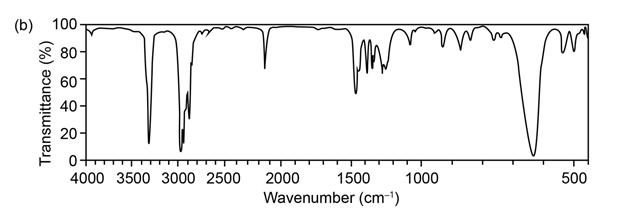
Question:The mass spectrum (a) and the infrared spectrum (b) of an unknown hydrocarbon are shown. Propose as many structures as you can.
The heat of hydrogenation for allene (Problem 7-61) to yield propane is -295 kJ/mol, and the heat of hydrogenation for a typical monosubstituted alkene, such as propene, is -125 kJ/mol. Is allene more stable or less stable than you might expect for a diene? Explain.
Which is stronger, the C=O bond in an ester () or the C=O bond in a saturated ketone ()? Explain
What do you think about this solution?
We value your feedback to improve our textbook solutions.