Chapter 12: 38E (page 385)
Which is stronger, the C=O bond in an ester () or the C=O bond in a saturated ketone ()? Explain
Short Answer
An ester C=O bondis stronger than a saturated ketone C=O bond.
Learning Materials
Features
Discover
Chapter 12: 38E (page 385)
Which is stronger, the C=O bond in an ester () or the C=O bond in a saturated ketone ()? Explain
An ester C=O bondis stronger than a saturated ketone C=O bond.
All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
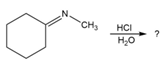
b.
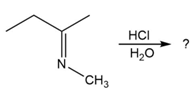
c.
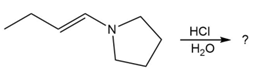
d.
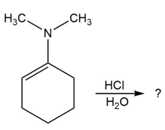
Answer questions 6-39 taking all stereoisomers into account.
(a)
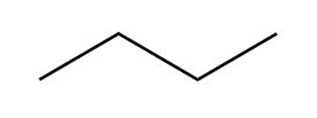
(b)
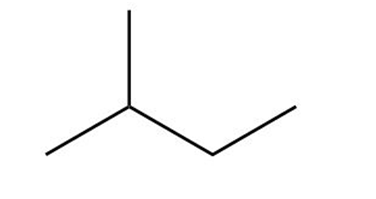
(c)
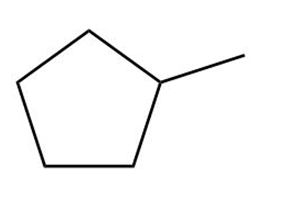
Question: Nitriles,, undergo a hydrolysis reaction when heated withaqueous acid. What is the structure of the compound produced by hydrolysis of propanenitrile, , if it has IR absorptions from and at , and has ?
Propose structures for compounds that meet the following descriptions:
Question: The infrared spectrum of the compound with the mass spectrum shown below lacks any significant absorption above . There is a prominent peak near and another strong peak near .Propose a structure consistent with the data.
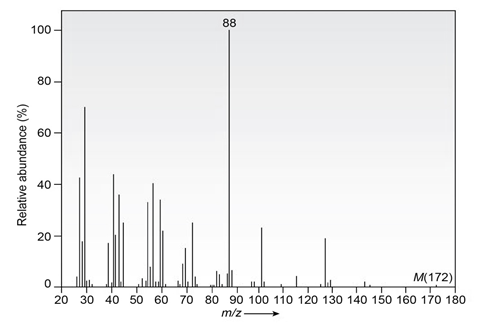
What do you think about this solution?
We value your feedback to improve our textbook solutions.