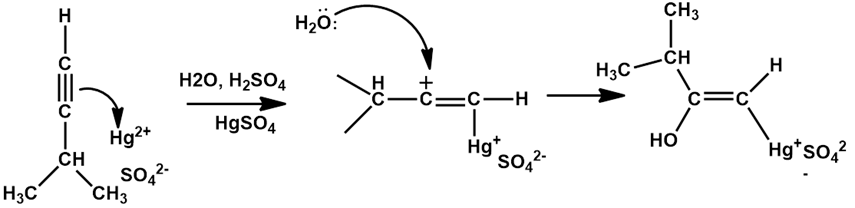
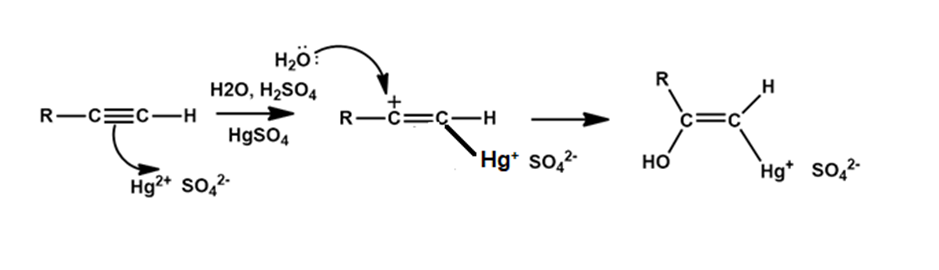
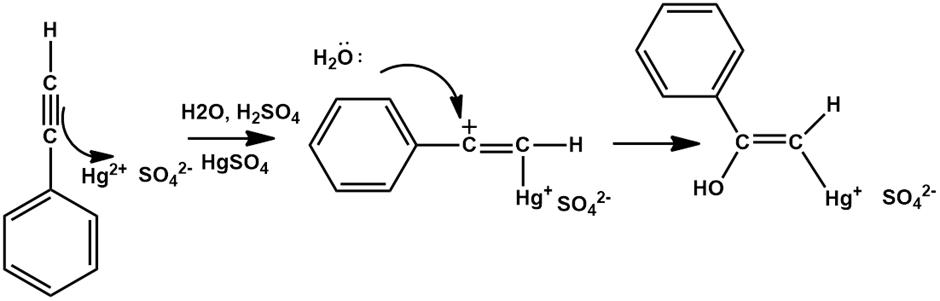
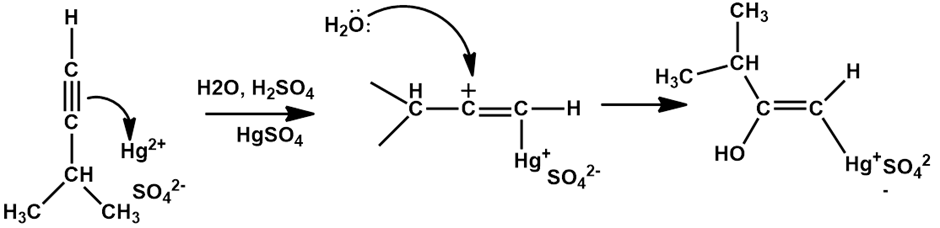
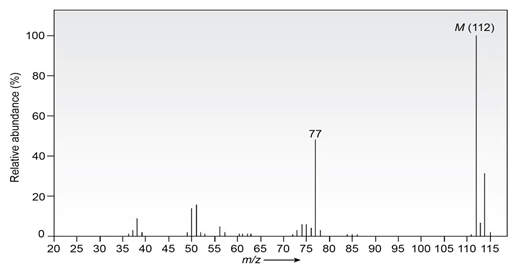
Chapter 12: Q20E (page 354)
The mercury-catalyzed hydration of alkynes involves the formation of an organomercury enol intermediate. Draw the electron-pushing mechanism to show how each of the following intermediates is formed.
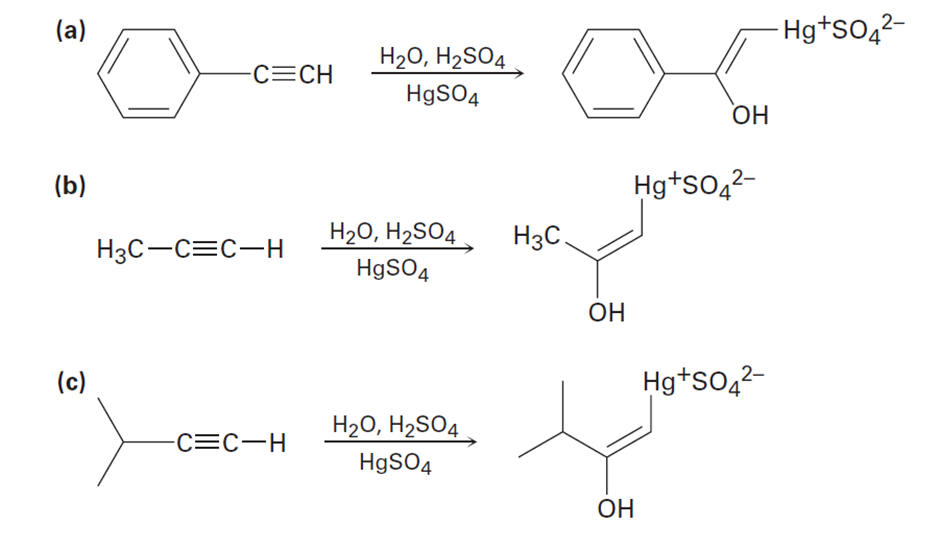
Short Answer
a).
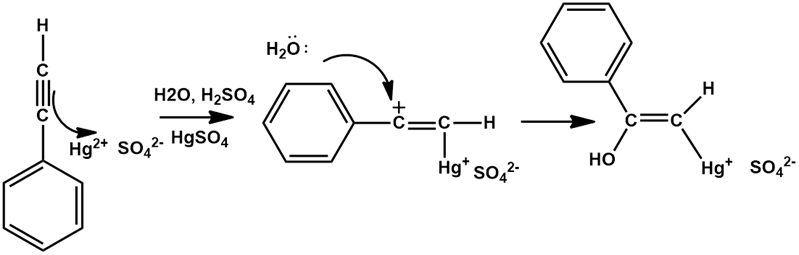
b).
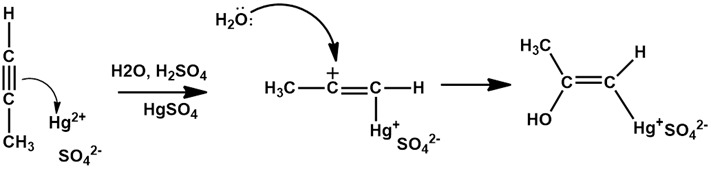
c).