Chapter 12: Q32E (page 354)
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
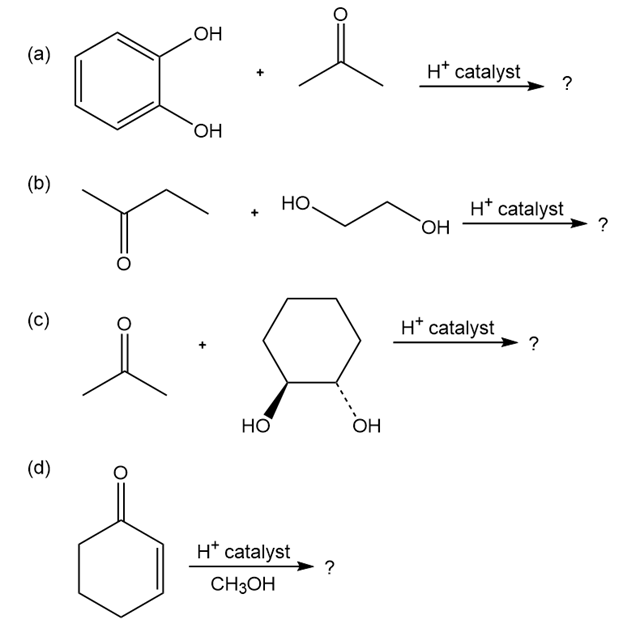
Short Answer
- The product of the first reaction is-
Learning Materials
Features
Discover
Chapter 12: Q32E (page 354)
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?


All the tools & learning materials you need for study success - in one app.
Get started for free
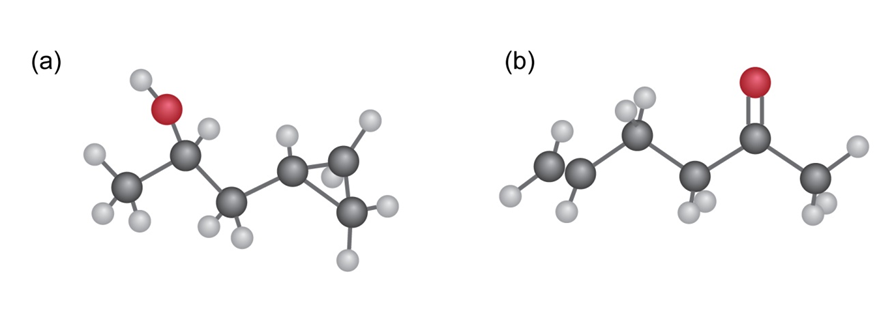
How might you use IR spectroscopy to distinguish between the followingpairs of isomers?
(a) CH3 CH2 OH and CH3 OCH3
(b) Cyclohexane and 1 - hexene
(c) CH3 CH2 CO2 H and HOCH2 CH2 CHO
Question: How could you use infrared spectroscopy to distinguish between the
following pairs of isomers?
(a)
(b)
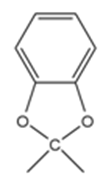
Grignard reagents undergo a general and very useful reaction with
ketones. Methylmagnesium bromide, for example, reacts with cyclohexanoneto yield a product with the formula C7H14O . What is the structure of this product if it has an IR absorption at 3400 cm-1 ?
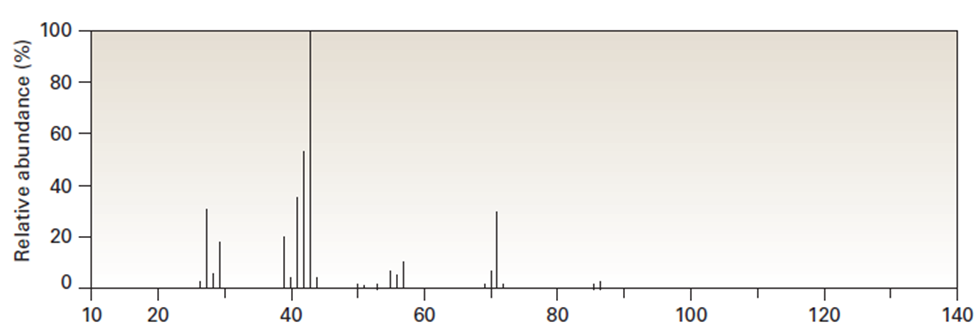
Question: 2-Methylpentane (C6H14)has the mass spectrum shown. Which peak
represents? Which is the base peak? Propose structures for fragment
ions of m/z=71, 57, 43, and 29. Why does the base peak have the mass it does?
How would you prepare the following substances, starting from any
compounds having four carbons or fewer?
What do you think about this solution?
We value your feedback to improve our textbook solutions.