Chapter 12: Q35c E (page 385)
Question: At what approximate positions might the following compounds show
IR absorptions?
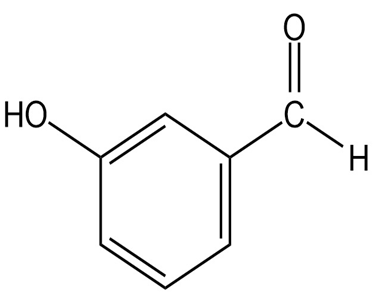
(c)
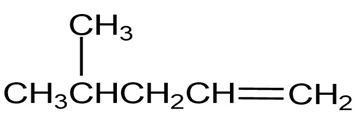
Short Answer
(c) It shows two distinguished absorptions due to at 910-990,
at 1640-1680 and at 3020-3100.
Learning Materials
Features
Discover
Chapter 12: Q35c E (page 385)
Question: At what approximate positions might the following compounds show
IR absorptions?
(c)

(c) It shows two distinguished absorptions due to at 910-990,
at 1640-1680 and at 3020-3100.
All the tools & learning materials you need for study success - in one app.
Get started for free
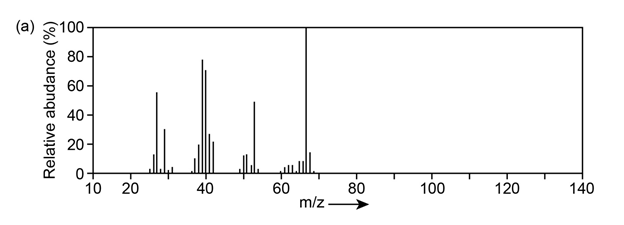
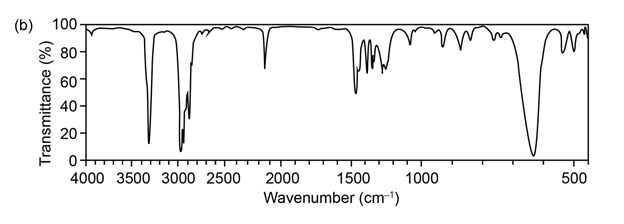
Question:The mass spectrum (a) and the infrared spectrum (b) of an unknown hydrocarbon are shown. Propose as many structures as you can.
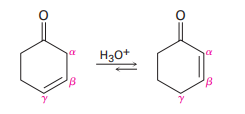
Nonconjugated ,-unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated ,-unsaturated isomers. Propose a mechanism for this isomerization.
Which of the following bases could be used to deprotonate 1-butyne?
(a) KOH
Question: Propose structures for compounds that meet the following descriptions:
A non-optically active compound with an IR absorption at.
Question: At what approximate positions might the following compounds showIR absorptions?
(f)
What do you think about this solution?
We value your feedback to improve our textbook solutions.