Chapter 2: 49E (page 59)
Calculate values from the following ’s:
(a)Acetone, =19.3 (b)Formic acid, =3.75
Short Answer
(a) The of Acetone isif its = 19.3.
(b) The of Formic acid isif its = 3.75.
Learning Materials
Features
Discover
Chapter 2: 49E (page 59)
Calculate values from the following ’s:
(a)Acetone, =19.3 (b)Formic acid, =3.75
(a) The of Acetone isif its = 19.3.
(b) The of Formic acid isif its = 3.75.
All the tools & learning materials you need for study success - in one app.
Get started for free
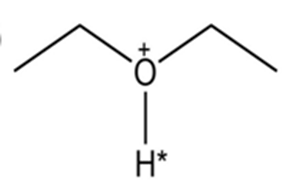
Draw the conjugate base for each compound below (the acidic hydrogen in each case is marked with an *).
(a)
Draw as many resonance structures as you can for the following species, adding appropriate formal charges to each:
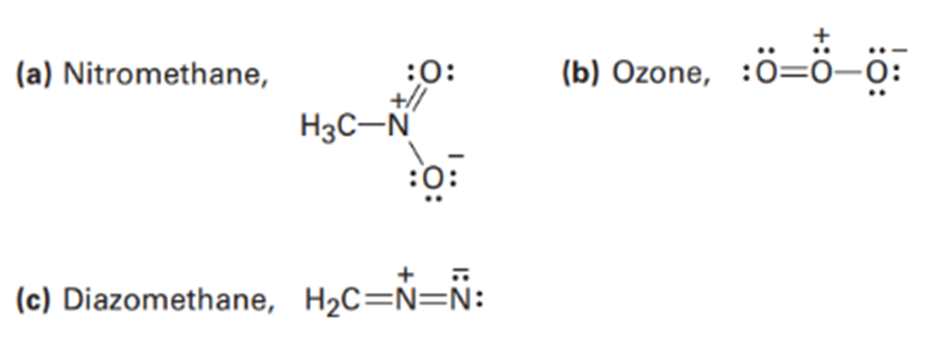
Rank the following substances in order of increasing acidity:
Use the table in Appendix B to determine in which direction the
Equilibrium is favored.
(b)
Ammonia, NH3, has=36, and acetone has=19. Will the following reaction take place to a significant extent?
What do you think about this solution?
We value your feedback to improve our textbook solutions.