Chapter 2: 50E (page 59)
Calculate values from the following ’s:
(a)Nitromethane, =
(b)Acrylic acid, =
Short Answer
(a) The value of Nitromethane is 10.30
(b) The value of acrylic acid is 4.25
Learning Materials
Features
Discover
Chapter 2: 50E (page 59)
Calculate values from the following ’s:
(a)Nitromethane, =
(b)Acrylic acid, =
(a) The value of Nitromethane is 10.30
(b) The value of acrylic acid is 4.25
All the tools & learning materials you need for study success - in one app.
Get started for free
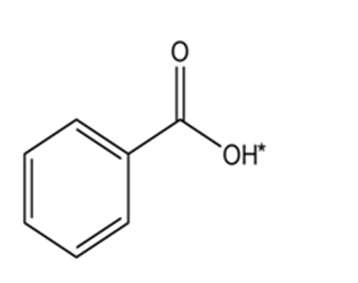
Draw the conjugate base for each compound below (the acidic hydrogen in each case is marked with an *).
(d)
Which intermolecular force is predominantly responsible for each
Observation below?
(a), a component found in paraffin wax, is a solid at
Room temperature while octane is a liquid.
(b) has a higher boiling point than
(c), which is found in vinegar, will dissolve in water but not
In oil—for simplicity you may assume oil is .
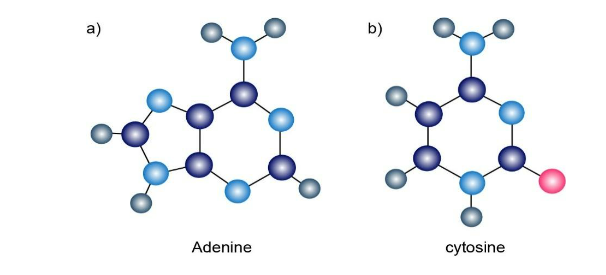
The following molecular models are representations of (a) adenine and (b) cytosine, constituents of DNA (deoxyribonucleic acid). Indicate the positions of multiple bonds and lone pairs for both, and draw skeletal structures (gray 5 C, pink 5 O, blue 5 N, ivory 5 H).
Assign formal charges to the atoms in each of the following molecules:
Question: Methanethiol, CH3SH , has a substantial dipole moment eventhough carbon and sulfur have identical electronegativities. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.