Chapter 2: Q. 2-2-66E-d (page 59)
Draw the conjugate base for each compound below (the acidic hydrogen in each case is marked with an *).
(d)
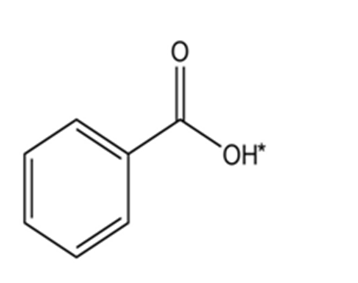
Short Answer
The conjugate base of the compound is:
Learning Materials
Features
Discover
Chapter 2: Q. 2-2-66E-d (page 59)
Draw the conjugate base for each compound below (the acidic hydrogen in each case is marked with an *).
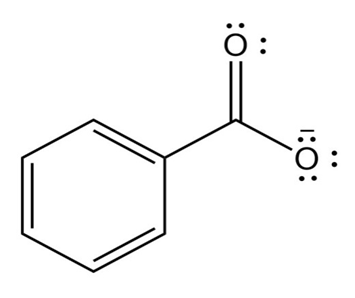
(d)

The conjugate base of the compound is:

All the tools & learning materials you need for study success - in one app.
Get started for free
Assign formal charges to the atoms in each of the following molecules:
a.
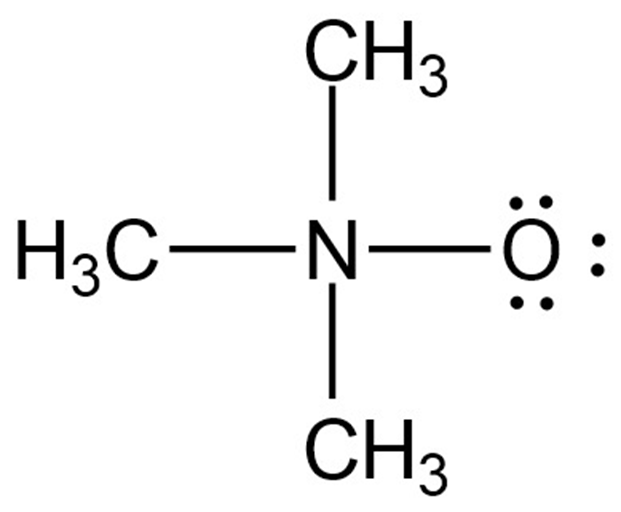
b.
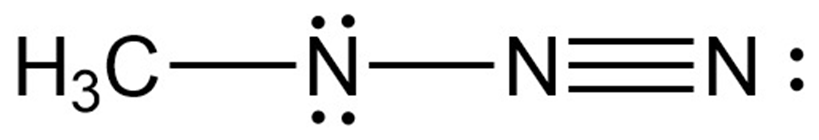
c.
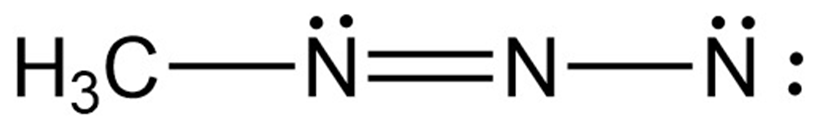
Use thed1/d2convention to indicate the direction of expected polarity for each of the bonds indicated.
(a)H3C–Cl (b)H3��–N��2(c)H2��–H
(d)H3C–SH (e)H3C–MgBr (f)H3��–F
Question: Assign formal charges to the atoms in each of the following molecules:
Will either of the following reactions take place to a significant extent as written, according to the data in Table 2-3?
a) b)
Determine if each compound or ion below has a dipole moment.
(a)Carbonate ion
What do you think about this solution?
We value your feedback to improve our textbook solutions.