Chapter 2: 63Ea (page 59)
Determine if each compound or ion below has a dipole moment.
(a)Carbonate ion
Short Answer
(a) Carbonate ion has no dipole moment.
Learning Materials
Features
Discover
Chapter 2: 63Ea (page 59)
Determine if each compound or ion below has a dipole moment.
(a)Carbonate ion
(a) Carbonate ion has no dipole moment.
All the tools & learning materials you need for study success - in one app.
Get started for free
Assign formal charges to the atoms in each of the following molecules:
Question: Use curved arrows to draw the protonated form of each Lewis base below.
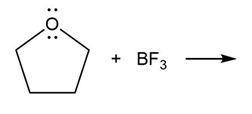
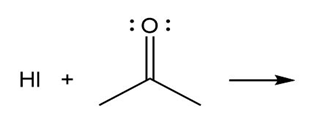
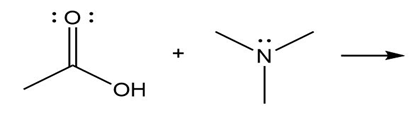
Question: Predict the product(s) of the acid/base reactions below. Draw curved arrows to show the formation and breaking of bonds.
a)
b)
c)
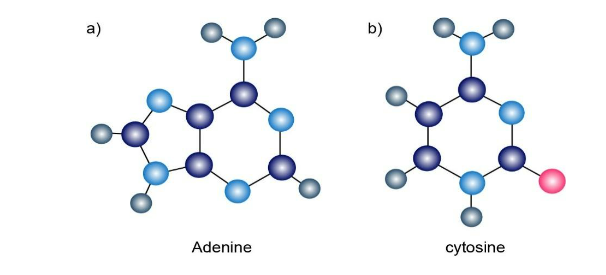
The following molecular models are representations of (a) adenine and (b) cytosine, constituents of DNA (deoxyribonucleic acid). Indicate the positions of multiple bonds and lone pairs for both, and draw skeletal structures (gray 5 C, pink 5 O, blue 5 N, ivory 5 H).
Use the electronegativity table given in Figure 2-2 to predict which bond in each of the following pairs is more polar, and indicate the direction of bond polarity for each compound.
a)or
b)or
c)or
d)or
What do you think about this solution?
We value your feedback to improve our textbook solutions.