Chapter 2: Q36E_a (page 59)
Assign formal charges to the atoms in each of the following molecules:
Short Answer
Answer
The formal charge on all H in is 0
The formal charge on C in is 0
The formal charge on O is -1
The formal charge on N is +1
Learning Materials
Features
Discover
Chapter 2: Q36E_a (page 59)
Assign formal charges to the atoms in each of the following molecules:
Answer
The formal charge on all H in is 0
The formal charge on C in is 0
The formal charge on O is -1
The formal charge on N is +1
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Phosgene, Cl2C=O , has a smaller dipole moment than formaldehyde,H2C=O , even though it contains electronegative chlorine atoms in place of hydrogen. Explain.
TheO-H hydrogen in acetic acid is more acidic than anyhydrogens. Explain this result using resonance structures.
Double bonds can also act like Lewis bases, sharing their electrons with Lewis acids. Use curved arrows to show how each double bond below will react with HCl and draw the resulting carbocation.
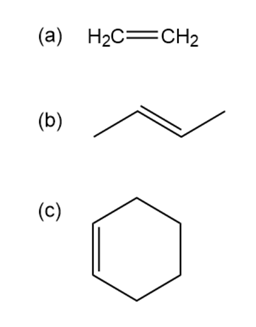
Identify the acids and bases in the following reactions:
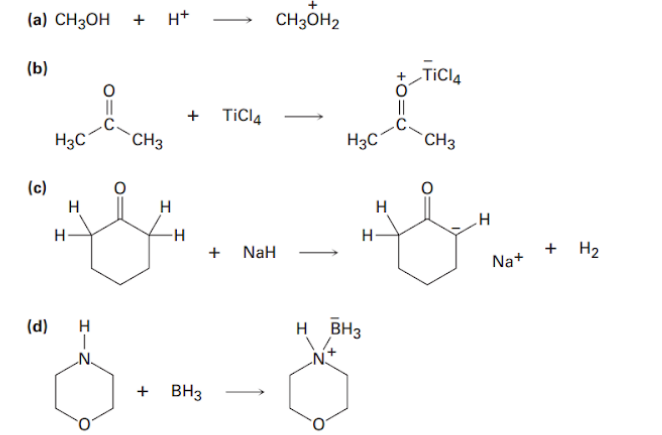
Which of the following molecules has a dipole moment? Indicate the expected direction of each.

What do you think about this solution?
We value your feedback to improve our textbook solutions.