Chapter 2: Q 2-2-41 E (page 59)
TheO-H hydrogen in acetic acid is more acidic than anyhydrogens. Explain this result using resonance structures.
Short Answer
OH hydrogen is resonance stabilized.
Learning Materials
Features
Discover
Chapter 2: Q 2-2-41 E (page 59)
TheO-H hydrogen in acetic acid is more acidic than anyhydrogens. Explain this result using resonance structures.
OH hydrogen is resonance stabilized.
All the tools & learning materials you need for study success - in one app.
Get started for free
Use the electronegativity values shown in Figure 2-2 to rank the following bonds from least polar to most polar: H3C–Li, H3C–K, H3C–F, H3C–MgBr, H3��–O��
Alcohols can act either as weak acids or as weak bases, just as water can. Show the reaction of methanol,, with a strong acid such as HCl and with a strong base such as.
Of the two vitamins A and C, one is hydrophilic and water-soluble while the other is hydrophobic and fat-soluble. Which is which?
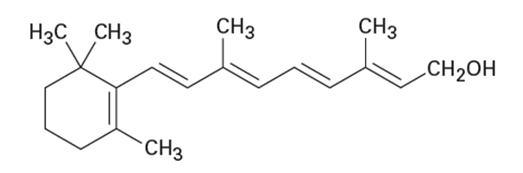
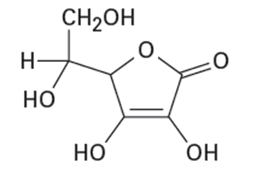
Vitamin A Vitamin C
(Retinol) (Ascorbic acid)
Question: Which of the following pairs of structures represent resonance forms?
Is tert-butoxide anion a strong enough base to react significantly with
water? In other words, can a solution of potassium tert-butoxide be
prepared in water? The pKa of tert-butyl alcohol is approximately 18.
What do you think about this solution?
We value your feedback to improve our textbook solutions.