Chapter 2: Q 2-2-40 E (page 59)
Alcohols can act either as weak acids or as weak bases, just as water can. Show the reaction of methanol,, with a strong acid such as HCl and with a strong base such as.
Short Answer
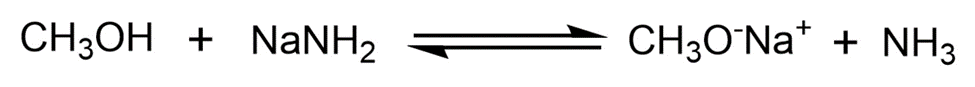
Learning Materials
Features
Discover
Chapter 2: Q 2-2-40 E (page 59)
Alcohols can act either as weak acids or as weak bases, just as water can. Show the reaction of methanol,, with a strong acid such as HCl and with a strong base such as.
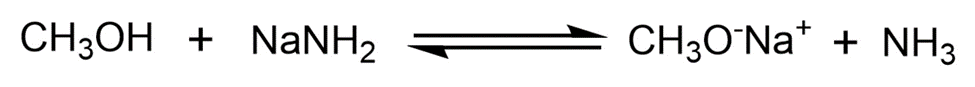
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw electron-dot structures for the following molecules, indicatingany unshared electron pairs. Which of the compounds are likely to actas Lewis acids and which as Lewis bases?
(a)(b) (c)
(d)HF (e)(f)
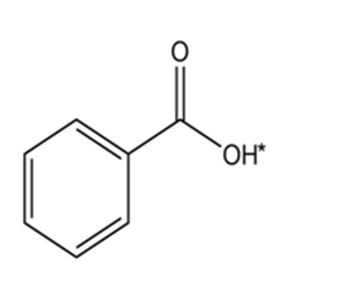
Draw the conjugate base for each compound below (the acidic hydrogen in each case is marked with an *).
(d)
We’ll see in the next chapter that organic molecules can be classified according to the functional groups they contain, where a functional group is a collection of atoms with a characteristic chemical reactivity. Use the electronegativity values given in Figure 2-2 on page 29 to predict the direction of polarization of the following functional groups.
Question:Phosgene, Cl2C=O , has a smaller dipole moment than formaldehyde,H2C=O , even though it contains electronegative chlorine atoms in place of hydrogen. Explain.
Question: Which of the following pairs of structures represent resonance forms, and which do not? Explain.
(a)
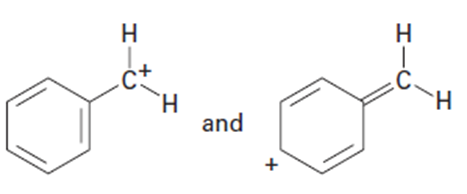
(b)
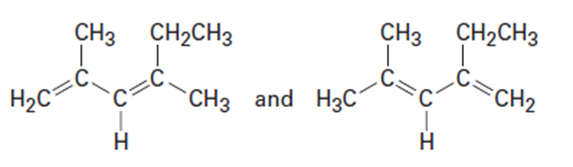
What do you think about this solution?
We value your feedback to improve our textbook solutions.