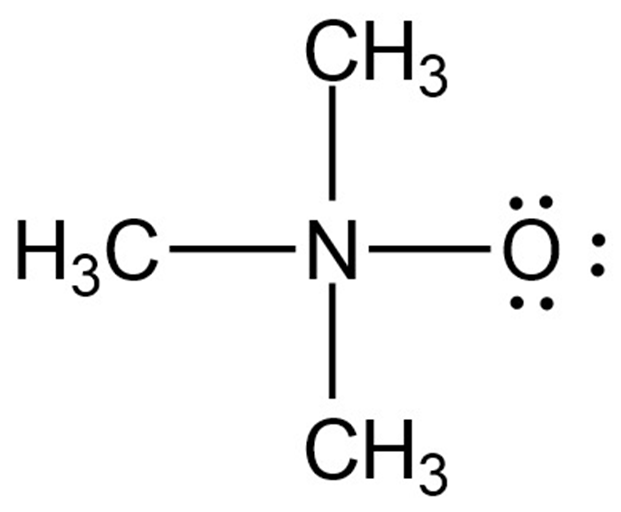
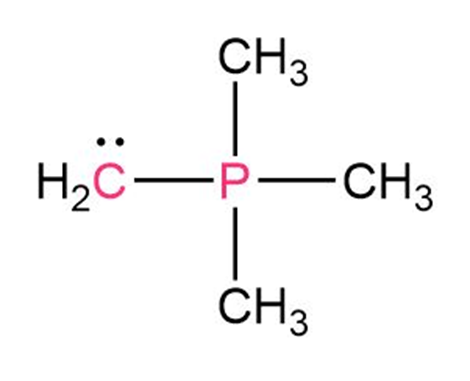
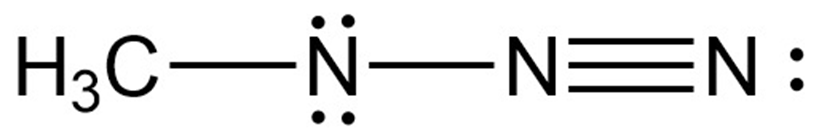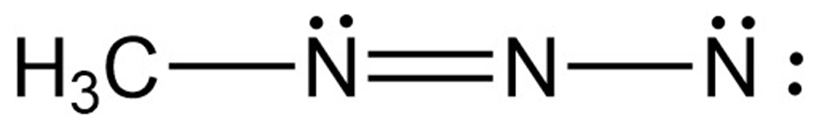Chapter 2: Q2-59E (page 59)
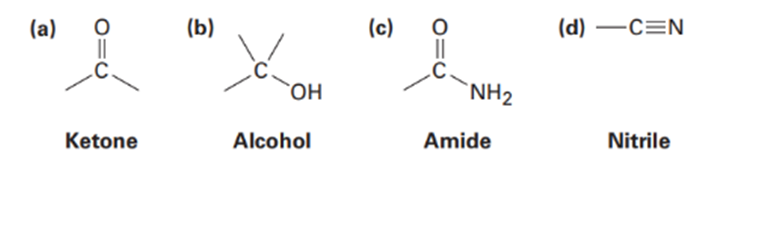
We’ll see in the next chapter that organic molecules can be classified according to the functional groups they contain, where a functional group is a collection of atoms with a characteristic chemical reactivity. Use the electronegativity values given in Figure 2-2 on page 29 to predict the direction of polarization of the following functional groups.
Short Answer
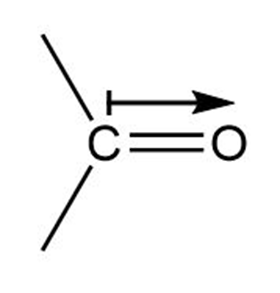
a. In ketone the direction of polarization from carbon to oxygen.
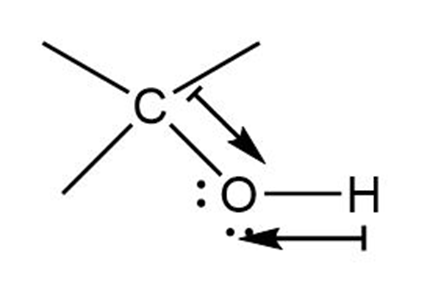
b. Direction of polarization from carbon to oxygen in Carbon-Oxygen bond.
Direction of Polarization from hydrogen to oxygen in Oxygen-hydrogen bond.
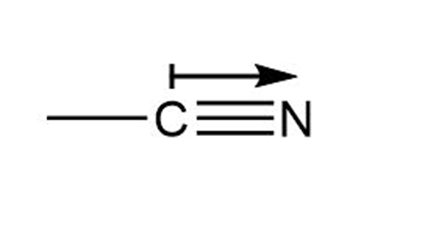
c. Direction of polarization from in Carbon-Oxygen bond.
Direction of Polarization from in Carbon-Nitrogen bond.
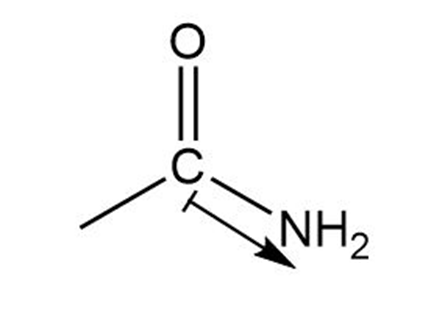
d. In ketone the direction of polarization from carbon to nitrogen.