Chapter 2: Q38E_b (page 59)
Question: Draw as many resonance structures as you can for the following
species:
Short Answer
Answer
These are three resonance forms.
Learning Materials
Features
Discover
Chapter 2: Q38E_b (page 59)
Question: Draw as many resonance structures as you can for the following
species:
Answer
These are three resonance forms.
All the tools & learning materials you need for study success - in one app.
Get started for free
Determine if each compound or ion below has a dipole moment.
(a)Carbonate ion
(b)
(c)
Draw as many resonance structures as you can for the following species, adding appropriate formal charges to each:
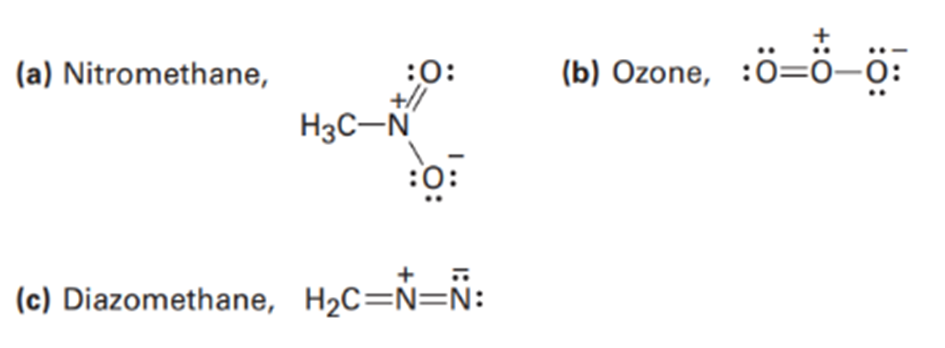
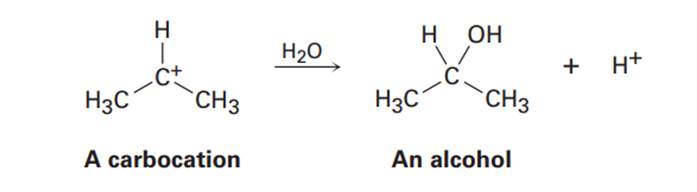
Carbocations, which contain a trivalent, positively charged carbon atom, react with water to give alcohols:
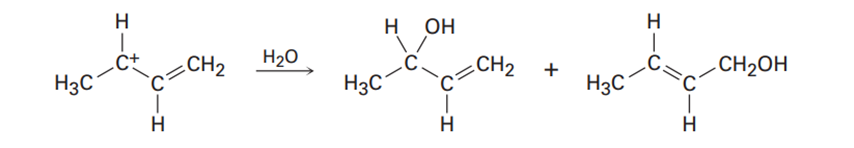
How can your account for the fact that the following carbocation gives a mixture of two alcohols in reaction with water?
Use the electronegativity table given in Figure 2-2 to predict which bond in each of the following pairs is more polar, and indicate the direction of bond polarity for each compound.
a)or
b)or
c)or
d)or
Imidazole forms part of the structure of the amino acid histidine and can act as both an acid and a base.
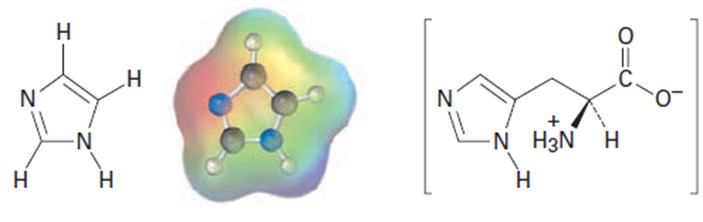
Imidazole Histidine
(a)Look at the electrostatic potential map of imidazole, and identify the most
acidic hydrogen atom and the most basic nitrogen atom.
(b)Draw structures for the resonance forms of the products that result when
imidazole is protonated by an acid and deprotonated by a base.
What do you think about this solution?
We value your feedback to improve our textbook solutions.