Chapter 2: Q38E_a (page 59)
Question: Draw as many resonance structures as you can for the following species.
Short Answer
Answer
These are two resonance forms.
Learning Materials
Features
Discover
Chapter 2: Q38E_a (page 59)
Question: Draw as many resonance structures as you can for the following species.
Answer
These are two resonance forms.
All the tools & learning materials you need for study success - in one app.
Get started for free
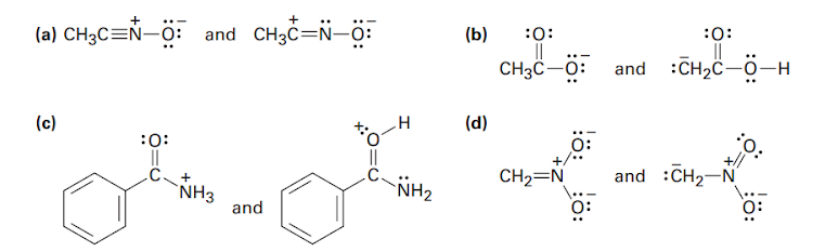
Which of the following pairs represent resonance structures?
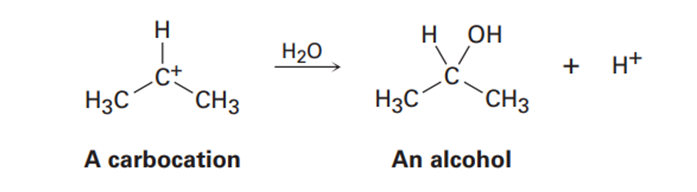
Carbocations, which contain a trivalent, positively charged carbon atom, react with water to give alcohols:
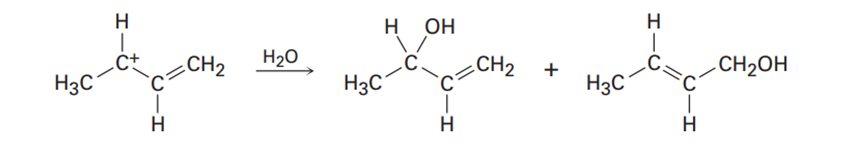
How can your account for the fact that the following carbocation gives a mixture of two alcohols in reaction with water?
Question: Calculate formal charges for the non-hydrogen atoms in the following molecules:
(a)Diazomethane,H2C=N=N:
(b)Acetonitrile oxide, H3C-CN-O
(c) Methyl isocyanides,H3CNC:
Question: Calculate the formal charges on the atoms shown in red.
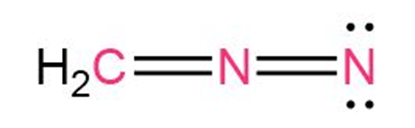
Question: Organic phosphate groups occur commonly in biological molecules. Calculate formal charges on the four O atoms in the methyl phosphate dianion.
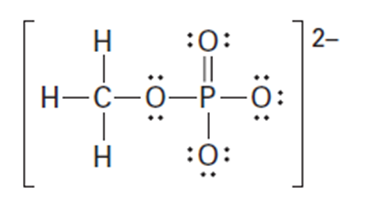
Methyl phosphate
What do you think about this solution?
We value your feedback to improve our textbook solutions.