Chapter 2: 63Eb (page 59)
Determine if each compound or ion below has a dipole moment.
(a)Carbonate ion
(b)
(c)
Short Answer
Dimethyl ether has a dipole moment.
Learning Materials
Features
Discover
Chapter 2: 63Eb (page 59)
Determine if each compound or ion below has a dipole moment.
(a)Carbonate ion
(b)
(c)
Dimethyl ether has a dipole moment.
All the tools & learning materials you need for study success - in one app.
Get started for free
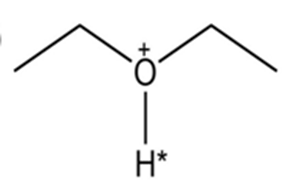
Draw the conjugate base for each compound below (the acidic hydrogen in each case is marked with an *).
(a)
Question: Calculate the formal charges on the atoms shown in red.
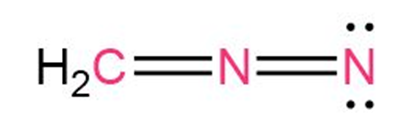
Use the electronegativity table given in Figure 2-2 to predict which bond in each of the following pairs is more polar, and indicate the direction of bond polarity for each compound.
a)or
b)or
c)or
d)or
Which, if any, of the substances in Problem 2-44 is a strong enough acidto react almost completely with NaOH? (The of is 15.74.)
Question: Which of the following pairs of structures represent resonance forms?
What do you think about this solution?
We value your feedback to improve our textbook solutions.