Chapter 2: Q. 2-2-66E-a (page 59)
Draw the conjugate base for each compound below (the acidic hydrogen in each case is marked with an *).
(a)
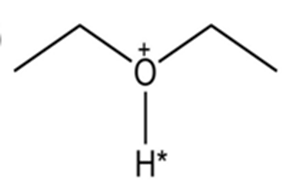
Short Answer
The conjugate base of the compound is:
Learning Materials
Features
Discover
Chapter 2: Q. 2-2-66E-a (page 59)
Draw the conjugate base for each compound below (the acidic hydrogen in each case is marked with an *).
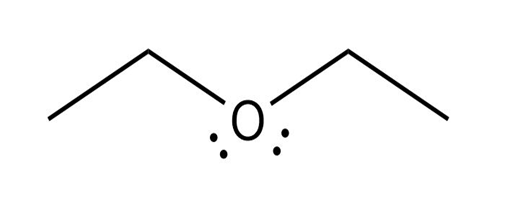
(a)

The conjugate base of the compound is:

All the tools & learning materials you need for study success - in one app.
Get started for free
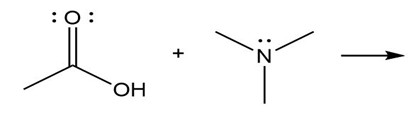
Question: Predict the product(s) of the acid/base reactions below. Draw curved arrows to show the formation and breaking of bonds.
a)
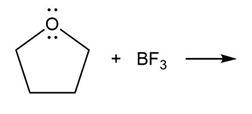
b)
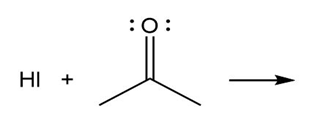
c)
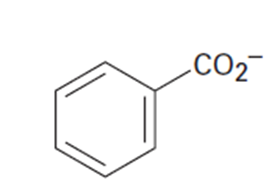
Draw the indicated number of resonance forms for each of the following
species:
(a)The methyl phosphate anion, CHOPO32-(3)
(b)The nitrate anion,NO3 (3)
(c)The allylcation,H2CPCHOCH2+(2)
(d)The benzoate anion (4)
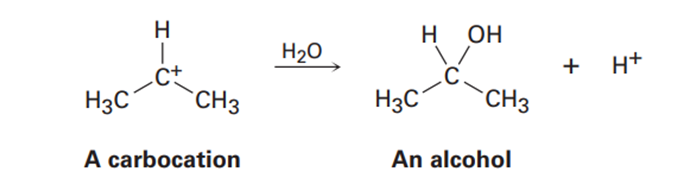
Carbocations, which contain a trivalent, positively charged carbon atom, react with water to give alcohols:
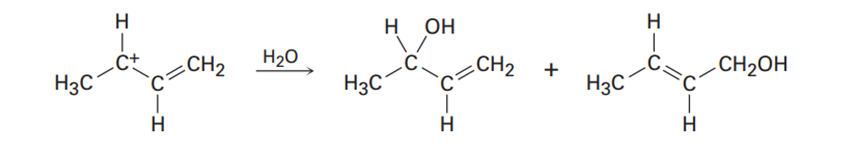
How can your account for the fact that the following carbocation gives a mixture of two alcohols in reaction with water?
Alcohols can act either as weak acids or as weak bases, just as water can. Show the reaction of methanol,, with a strong acid such as HCl and with a strong base such as.
Question: Nitric acid (HNO3) reacts with ammonia (NH3) to yield ammonium nitrate. Write the reaction, and identify the acid, the base, the conjugate acid product, and the conjugate base product.
What do you think about this solution?
We value your feedback to improve our textbook solutions.