Chapter 2: Q 24 E (page 59)
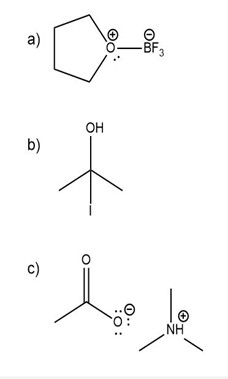
Question: Predict the product(s) of the acid/base reactions below. Draw curved arrows to show the formation and breaking of bonds.
a)
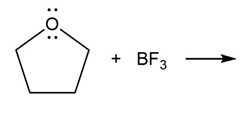
b)
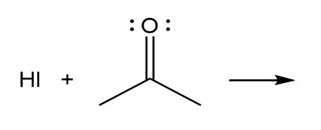
c)
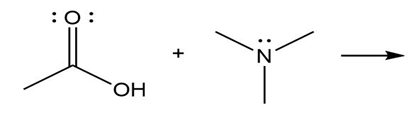
Short Answer
Learning Materials
Features
Discover
Chapter 2: Q 24 E (page 59)
Question: Predict the product(s) of the acid/base reactions below. Draw curved arrows to show the formation and breaking of bonds.
a)

b)

c)


All the tools & learning materials you need for study success - in one app.
Get started for free
Of the two vitamins A and C, one is hydrophilic and water-soluble while the other is hydrophobic and fat-soluble. Which is which?
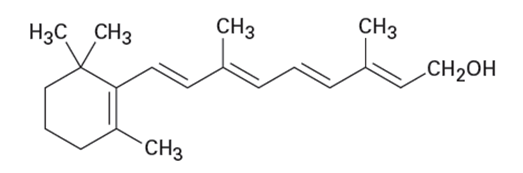
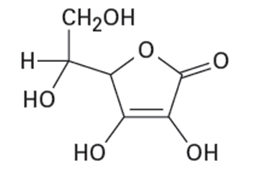
Vitamin A Vitamin C
(Retinol) (Ascorbic acid)
Imidazole forms part of the structure of the amino acid histidine and can act as both an acid and a base.
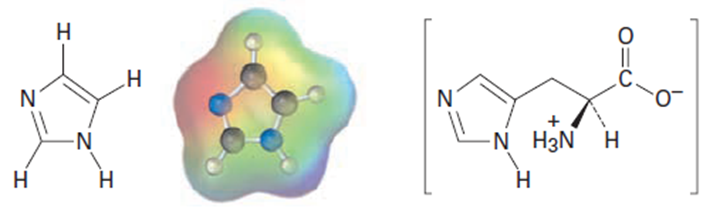
Imidazole Histidine
(a)Look at the electrostatic potential map of imidazole, and identify the most
acidic hydrogen atom and the most basic nitrogen atom.
(b)Draw structures for the resonance forms of the products that result when
imidazole is protonated by an acid and deprotonated by a base.
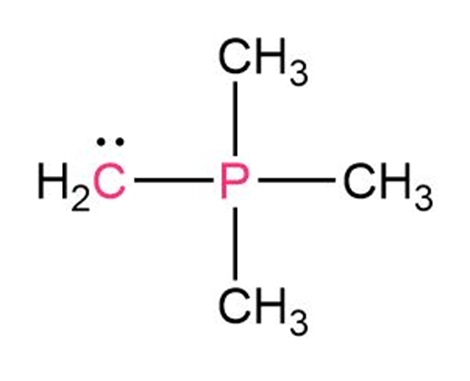
Question: Draw as many resonance structures as you can for the following
species:
Question: Calculate the formal charges on the atoms shown in red.
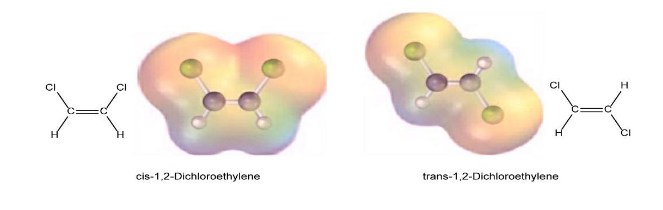
Question: Cis-1, 2-Dichloroethylene, and trans-1, 2-dichloroethylene are isomers, compounds with the same formula but different chemical structures. Look at the following electrostatic potential maps, and tell whether either compound has a dipole moment.
What do you think about this solution?
We value your feedback to improve our textbook solutions.