Chapter 2: Q. 2-2-64E-c (page 59)
Use the table in Appendix B to determine in which direction the
Equilibrium is favored.
(c)localid="1650537382286"
Short Answer
Equilibrium favors the direction of the product.
Learning Materials
Features
Discover
Chapter 2: Q. 2-2-64E-c (page 59)
Use the table in Appendix B to determine in which direction the
Equilibrium is favored.
(c)localid="1650537382286"
Equilibrium favors the direction of the product.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Phosgene, Cl2C=O , has a smaller dipole moment than formaldehyde, H2C=O , even though it contains electronegative chlorine atoms in place of hydrogen. Explain.
TheO-H hydrogen in acetic acid is more acidic than anyhydrogens. Explain this result using resonance structures.
Predict the structure of the product formed in the reaction of the organic base pyridine with the organic acid acetic acid, and use curved arrowsto indicate the direction of electron flow.
Rank the following substances in order of increasing acidity:
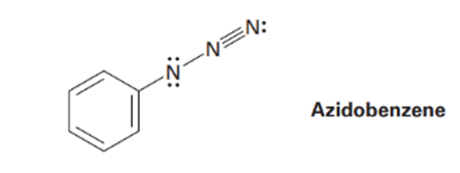
The azide functional group, which occurs in azido benzene, contains three adjacent nitrogen atoms. One resonance structure for azido benzene is shown. Draw three additional resonance structures, and assign appropriate formal charges to the atoms in all four.
What do you think about this solution?
We value your feedback to improve our textbook solutions.