Chapter 2: 44E (page 59)
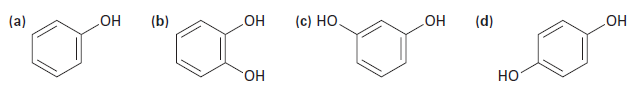
Rank the following substances in order of increasing acidity:
Short Answer
The substance with the largest value of pKa is the least acidic. So, the order of increasing acidity is:
Learning Materials
Features
Discover
Chapter 2: 44E (page 59)
Rank the following substances in order of increasing acidity:
The substance with the largest value of pKa is the least acidic. So, the order of increasing acidity is:
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:
(A)The bond length is . What would the dipole moment of be if the molecule were ionic, ?
(B)The actual dipole moment of HCl is . What is the percent
ionic character of the bond?
Question: Organic phosphate groups occur commonly in biological molecules. Calculate formal charges on the four O atoms in the methyl phosphate dianion.
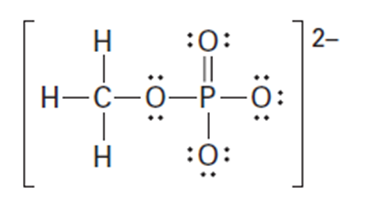
Methyl phosphate
Write the products of the following acid–base reactions:
Predict the structure of the product formed in the reaction of the organic base pyridine with the organic acid acetic acid, and use curved arrowsto indicate the direction of electron flow.
Which of the following molecules has a dipole moment? Indicate the expected direction of each.
What do you think about this solution?
We value your feedback to improve our textbook solutions.