Chapter 2: 43E (page 59)
Write the products of the following acid–base reactions:
Short Answer
The products of the following acid-base reaction are given below:
Learning Materials
Features
Discover
Chapter 2: 43E (page 59)
Write the products of the following acid–base reactions:
The products of the following acid-base reaction are given below:
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which of the following pairs of structures represent resonance forms?
Is tert-butoxide anion a strong enough base to react significantly with
water? In other words, can a solution of potassium tert-butoxide be
prepared in water? The pKa of tert-butyl alcohol is approximately 18.
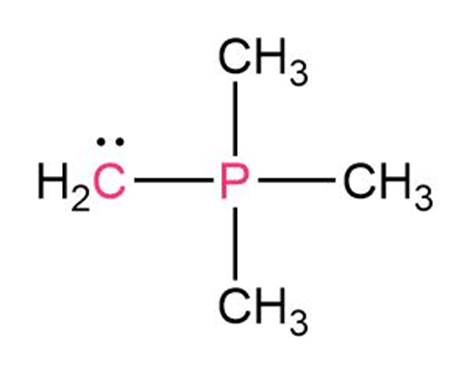
Draw electron-dot structures for the following molecules, indicatingany unshared electron pairs. Which of the compounds are likely to actas Lewis acids and which as Lewis bases?
(a)(b) (c)
(d)HF (e)(f)
Use the electronegativity values shown in Figure 2-2 to rank the following bonds from least polar to most polar: H3C–Li, H3C–K, H3C–F, H3C–MgBr, H3��–O��
Question: Calculate the formal charges on the atoms shown in red.
What do you think about this solution?
We value your feedback to improve our textbook solutions.