Chapter 2: Q37E_b (page 59)
Question: Which of the following pairs of structures represent resonance forms?
Short Answer
Answer
These are resonance forms.
Learning Materials
Features
Discover
Chapter 2: Q37E_b (page 59)
Question: Which of the following pairs of structures represent resonance forms?
Answer
These are resonance forms.
All the tools & learning materials you need for study success - in one app.
Get started for free
Sodium bicarbonate, NaHCO3, is the sodium salt of carbonic acid
, =6.37. Which of the substances shown in Problem 2-44
will react significantly with sodium bicarbonate?
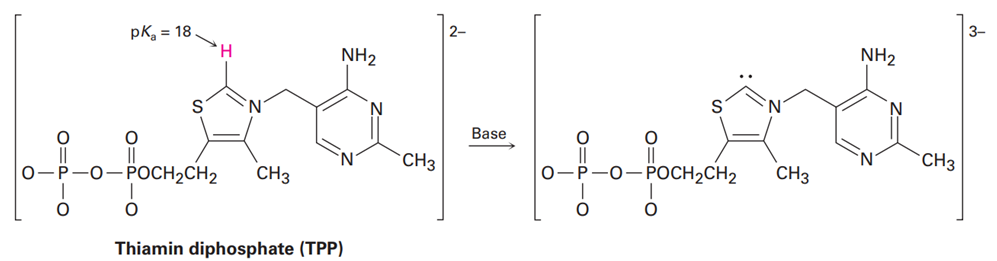
Thiamin diphosphate (TPP), a derivative of vitamin B1 required for glucose metabolism, is a weak acid that can be deprotonated by a base. Assign formal charges to the appropriate atoms in both TPP and its deprotonation product.
Ethylene glycol,, may look nonpolar when drawn, but an
internal hydrogen bond results in an electric dipole moment. Explain.
Use the table in Appendix B to determine in which direction the
Equilibrium is favored.
(b)
Will either of the following reactions take place to a significant extent as written, according to the data in Table 2-3?
a) b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.