Chapter 2: Q38E_c (page 59)
Question: Draw as many resonance structures as you can for the following
species:
Short Answer
Answer
These are three resonance forms.
Learning Materials
Features
Discover
Chapter 2: Q38E_c (page 59)
Question: Draw as many resonance structures as you can for the following
species:
Answer
These are three resonance forms.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Use curved arrows to draw the protonated form of each Lewis base below.
Question: Assign formal charges to the atoms in each of the following molecules:
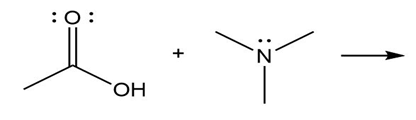
Question: Predict the product(s) of the acid/base reactions below. Draw curved arrows to show the formation and breaking of bonds.
a)
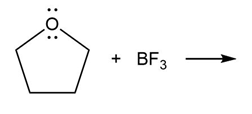
b)
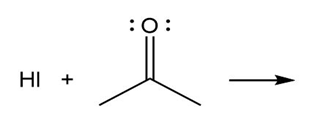
c)
1,1,1-Trichloroethanol is an acid more than 1000 times stronger thanethanol, even though both have a conjugate base where the negativecharge is on an oxygen. Provide an explanation for this observation.
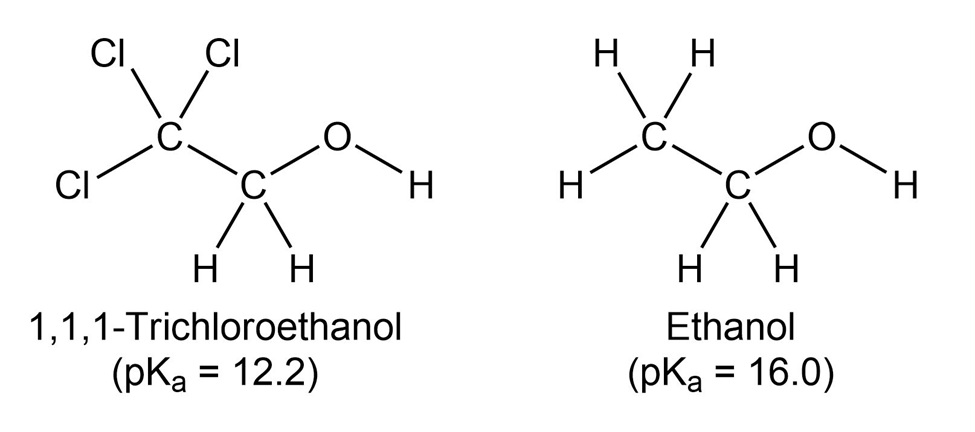
Assume that you have two unlabeled bottles, one of which contains
phenol ( 9.9) and one of which contains acetic acid (=4.76).
In light of your answer to Problem 2-52, suggest a simple way to determine
what is in each bottle.
What do you think about this solution?
We value your feedback to improve our textbook solutions.