Chapter 2: Q36E_c (page 59)
Question: Assign formal charges to the atoms in each of the following molecules:
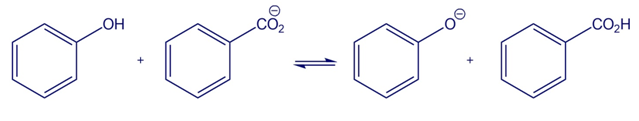
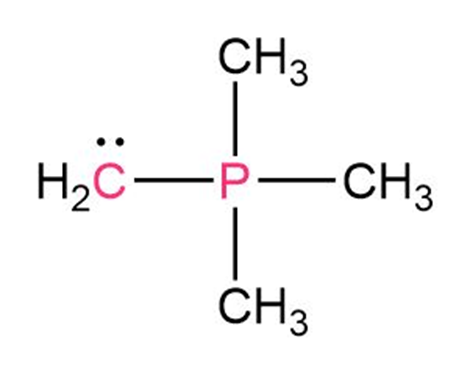
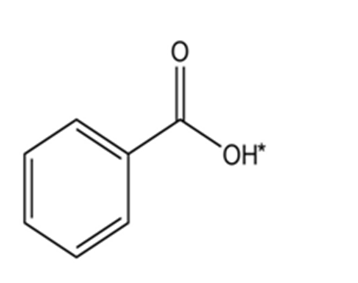
Short Answer
Answer
The formal charge on all H in is 0.
The formal charge on C in is 0.
The formal charge on N at left is 0.
The formal charge on N in the middle is +1.
The formal charge on N at the rightmost is -1.