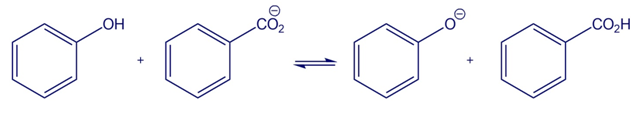
Chapter 2: Q64E-a (page 59)
Use the pKa table in Appendix B to determine in which direction the
Equilibrium is favored.
(a)
Short Answer
A reactant is favoured in this reaction
Learning Materials
Features
Discover
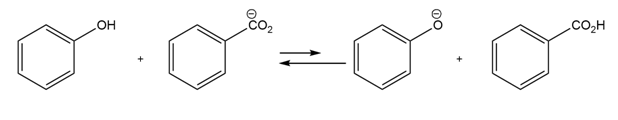
Chapter 2: Q64E-a (page 59)
Use the pKa table in Appendix B to determine in which direction the
Equilibrium is favored.
(a)

A reactant is favoured in this reaction

All the tools & learning materials you need for study success - in one app.
Get started for free
Use thed1/d2convention to indicate the direction of expected polarity for each of the bonds indicated.
(a)H3C–Cl (b)H3��–N��2(c)H2��–H
(d)H3C–SH (e)H3C–MgBr (f)H3��–F
Question: Which of the following pairs of structures represent resonance forms?
Determine if each compound or ion below has a dipole moment.
(a)Carbonate ion
(b)
(c)
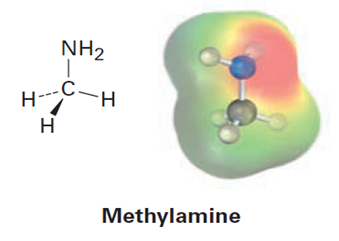
Look at the following electrostatic potential map of methylamine, a substance responsible for the odor of rotting fish, and tell the direction of polarization of the C-N bond:
Which intermolecular force is predominantly responsible for each
Observation below?
(a), a component found in paraffin wax, is a solid at
Room temperature while octane is a liquid.
(b) has a higher boiling point than
(c), which is found in vinegar, will dissolve in water but not
In oil—for simplicity you may assume oil is .
What do you think about this solution?
We value your feedback to improve our textbook solutions.