Chapter 2: Q37E_d (page 59)
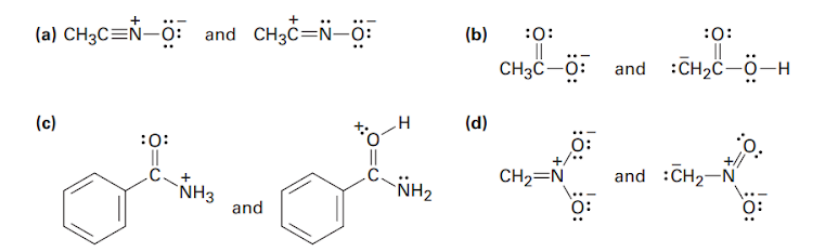
Question: Which of the following pairs of structures represent resonance forms?
Short Answer
Answer
These are resonance forms.
Learning Materials
Features
Discover
Chapter 2: Q37E_d (page 59)
Question: Which of the following pairs of structures represent resonance forms?
Answer
These are resonance forms.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Assign formal charges to the atoms in each of the following molecules:
Draw electron-dot structures for the following molecules, indicatingany unshared electron pairs. Which of the compounds are likely to actas Lewis acids and which as Lewis bases?
(a)(b) (c)
(d)HF (e)(f)
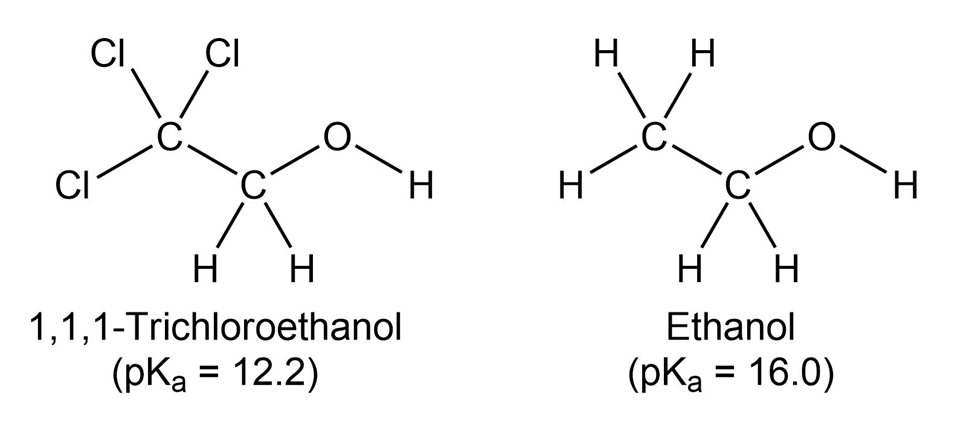
1,1,1-Trichloroethanol is an acid more than 1000 times stronger thanethanol, even though both have a conjugate base where the negativecharge is on an oxygen. Provide an explanation for this observation.
Which of the following pairs represent resonance structures?
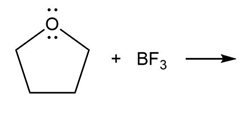
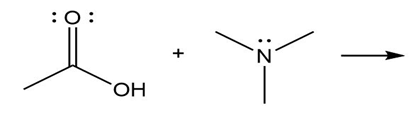
Question: Predict the product(s) of the acid/base reactions below. Draw curved arrows to show the formation and breaking of bonds.
a)
b)
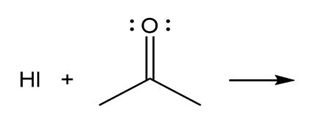
c)
What do you think about this solution?
We value your feedback to improve our textbook solutions.